用于全合成 simnotrelvir 的经典肽偶联方法
IF 2.1
3区 化学
Q2 CHEMISTRY, ORGANIC
引用次数: 0
摘要
我们完成了口服小分子抗病毒剂的全合成;这是一种基于结构的开发药物 simnotrelvir。该合成涉及容易获得的商用氨基酸 L-叔亮氨酸、4-羟基-l-脯氨酸和 l-谷氨酸,它们很容易转化为关键的中间体,并通过使用 HATU 和 NMM 的经典肽偶联技术偶联在一起。合成过程中的一个关键步骤是在 ZnCl2 的介导下对叔亮氨酸氨基酸分子进行 Boc-脱保护和三氟乙酰化反应。本文章由计算机程序翻译,如有差异,请以英文原文为准。
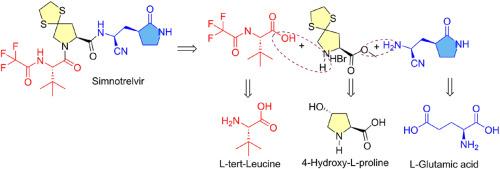
A classical peptide coupling approach for the total synthesis of simnotrelvir
The total synthesis of an oral small-molecule antiviral agent; a structure based developmental drug simnotrelvir has been accomplished. The synthesis involves readily accessible commercial amino acids L-tert-leucine, 4-hydroxy-l-proline and l-glutamic acid that are easily converted to the key intermediates and are coupled together following classical peptide coupling technique using HATU and NMM. One of the crucial step in the synthesis involved a one pot Boc-deprotection and trifluoroacetylation of tert-leucine amino acid moiety mediated by ZnCl2.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Tetrahedron
化学-有机化学
CiteScore
3.90
自引率
4.80%
发文量
439
审稿时长
34 days
期刊介绍:
Tetrahedron publishes full accounts of research having outstanding significance in the broad field of organic chemistry and its related disciplines, such as organic materials and bio-organic chemistry.
Regular papers in Tetrahedron are expected to represent detailed accounts of an original study having substantially greater scope and details than that found in a communication, as published in Tetrahedron Letters.
Tetrahedron also publishes thematic collections of papers as special issues and ''Reports'', commissioned in-depth reviews providing a comprehensive overview of a research area.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


