小鼠线粒体 ND5 无义突变的综合表型评估。
IF 9.5
2区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
线粒体 DNA(mtDNA)突变引起的线粒体功能障碍与多种人类疾病有关。要对线粒体遗传疾病进行全面分析,就必须为人类疾病研究建立合适的动物模型。虽然通过过早终止密码子敲除基因是研究目标基因独特功能的有效方法,但实现 mtDNA 基因敲除的方法还很少见。在这里,我们报告了异质MT-ND5基因敲除小鼠的基因型和表型。这些突变小鼠大脑皮层线粒体嵴受损、海马萎缩和不对称,导致学习和记忆异常。此外,突变小鼠易患肥胖症和发热性疾病。我们认为,这些 mtDNA 基因敲除小鼠可作为研究 MT-ND5 基因和开发人类线粒体疾病疗法的宝贵动物模型。本文章由计算机程序翻译,如有差异,请以英文原文为准。
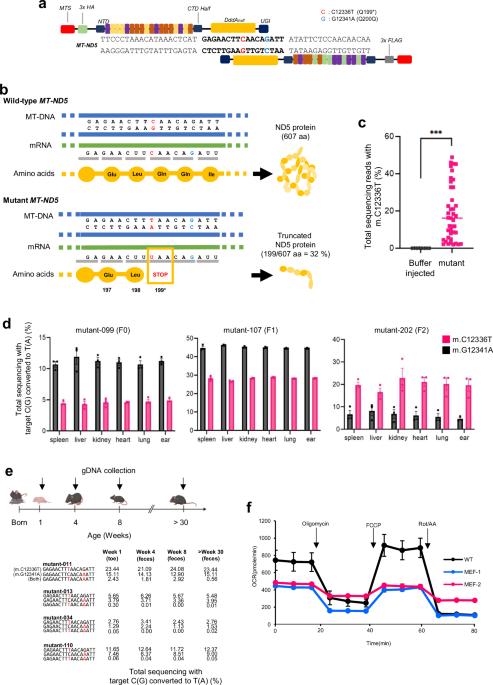
Comprehensive phenotypic assessment of nonsense mutations in mitochondrial ND5 in mice
Mitochondrial dysfunction induced by mitochondrial DNA (mtDNA) mutations has been implicated in various human diseases. A comprehensive analysis of mitochondrial genetic disorders requires suitable animal models for human disease studies. While gene knockout via premature stop codons is a powerful method for investigating the unique functions of target genes, achieving knockout of mtDNA has been rare. Here, we report the genotypes and phenotypes of heteroplasmic MT-ND5 gene-knockout mice. These mutant mice presented damaged mitochondrial cristae in the cerebral cortex, hippocampal atrophy, and asymmetry, leading to learning and memory abnormalities. Moreover, mutant mice are susceptible to obesity and thermogenetic disorders. We propose that these mtDNA gene-knockdown mice could serve as valuable animal models for studying the MT-ND5 gene and developing therapies for human mitochondrial disorders in the future. Mitochondria, often referred to as cell''s powerhouses, are essential for energy production and health. In this study, researchers used recently developed ways to edit mtDNA, allowing precise changes to study disease results in animal models. They performed an experiment involving the injection of specially designed mRNAs, into mouse embryos to cause specific changes in mtDNA. This method allowed the creation of mice with targeted mitochondrial gene removals, providing a useful model for studying mitochondrial diseases. Researchers found that mice with certain mtDNA changes showed symptoms like human mitochondrial diseases, such as brain abnormalities and metabolic issues. These findings emphasize the importance of mtDNA in health and disease, providing insights into how specific changes can affect bodily functions. Future research may use these findings to develop therapies targeting mtDNA changes. This summary was initially drafted using artificial intelligence, then revised and fact-checked by the author.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Experimental and Molecular Medicine
医学-生化与分子生物学
CiteScore
19.50
自引率
0.80%
发文量
166
审稿时长
3 months
期刊介绍:
Experimental & Molecular Medicine (EMM) stands as Korea's pioneering biochemistry journal, established in 1964 and rejuvenated in 1996 as an Open Access, fully peer-reviewed international journal. Dedicated to advancing translational research and showcasing recent breakthroughs in the biomedical realm, EMM invites submissions encompassing genetic, molecular, and cellular studies of human physiology and diseases. Emphasizing the correlation between experimental and translational research and enhanced clinical benefits, the journal actively encourages contributions employing specific molecular tools. Welcoming studies that bridge basic discoveries with clinical relevance, alongside articles demonstrating clear in vivo significance and novelty, Experimental & Molecular Medicine proudly serves as an open-access, online-only repository of cutting-edge medical research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


