依赖 SLC12A9 的离子转运机制维持溶酶体渗透压
IF 10.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
氨是一种无处不在的有毒细胞代谢副产物。氨的高膜渗透性和质子亲和性导致氨以膜渗透性差的形式:铵(NH4+)积聚在酸性溶酶体内。氨的积累会损害溶酶体的功能,这表明存在保护细胞免受氨毒性的机制。在这里,我们发现 SLC12A9 是一种溶酶体驻留蛋白,它通过控制氨和氯的水平来维持细胞器的平衡。SLC12A9 基因敲除(KO)细胞表现出溶酶体明显增大和铵含量升高。在移除铵的代谢源或溶酶体 pH 梯度消失后,这些表型被逆转。在 SLC12A9 KO 细胞中,溶酶体氯化物增加,铵转运需要 SLC12A9 与氯化物结合。我们的数据表明,SLC12A9 的功能是处理溶酶体氨和氯化物的核心,这是一种未被重视的溶酶体生理基本机制,可能与氨升高的组织(如肿瘤)特别相关。本文章由计算机程序翻译,如有差异,请以英文原文为准。
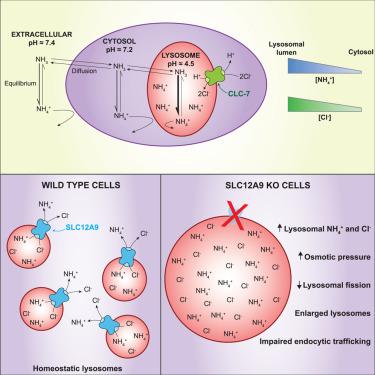
An SLC12A9-dependent ion transport mechanism maintains lysosomal osmolarity
Ammonia is a ubiquitous, toxic by-product of cell metabolism. Its high membrane permeability and proton affinity cause ammonia to accumulate inside acidic lysosomes in its poorly membrane-permeant form: ammonium (NH4+). Ammonium buildup compromises lysosomal function, suggesting the existence of mechanisms that protect cells from ammonium toxicity. Here, we identified SLC12A9 as a lysosomal-resident protein that preserves organelle homeostasis by controlling ammonium and chloride levels. SLC12A9 knockout (KO) cells showed grossly enlarged lysosomes and elevated ammonium content. These phenotypes were reversed upon removal of the metabolic source of ammonium or dissipation of the lysosomal pH gradient. Lysosomal chloride increased in SLC12A9 KO cells, and chloride binding by SLC12A9 was required for ammonium transport. Our data indicate that SLC12A9 function is central for the handling of lysosomal ammonium and chloride, an unappreciated, fundamental mechanism of lysosomal physiology that may have special relevance in tissues with elevated ammonia, such as tumors.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Developmental cell
生物-发育生物学
CiteScore
18.90
自引率
1.70%
发文量
203
审稿时长
3-6 weeks
期刊介绍:
Developmental Cell, established in 2001, is a comprehensive journal that explores a wide range of topics in cell and developmental biology. Our publication encompasses work across various disciplines within biology, with a particular emphasis on investigating the intersections between cell biology, developmental biology, and other related fields. Our primary objective is to present research conducted through a cell biological perspective, addressing the essential mechanisms governing cell function, cellular interactions, and responses to the environment. Moreover, we focus on understanding the collective behavior of cells, culminating in the formation of tissues, organs, and whole organisms, while also investigating the consequences of any malfunctions in these intricate processes.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


