通过靶向 IKKβ/NF-κB 信号传递,受控 TPCA-1 递送可设计出有利于肌腱再生的增生龛位
IF 18
1区 医学
Q1 ENGINEERING, BIOMEDICAL
引用次数: 0
摘要
由于肌腱的内在愈合能力较差,肌腱修复仍具有挑战性,而干细胞疗法已成为促进肌腱再生的一种有前途的策略。然而,急性肌腱损伤后的炎症环境破坏了干细胞分化,导致结果不尽人意。我们的研究通过多组学分析认识到,NF-κB信号在急性肌腱损伤后激活炎症和抑制干细胞韧化分化中起着关键作用。TPCA-1是IKKβ/NF-κB信号传导的选择性抑制剂,它能有效恢复炎症环境中受损的干细胞韧化分化。通过开发一种用于干细胞递送和TPCA-1控释的微球包合水凝胶系统,我们成功地设计了一个有利于肌腱再生的韧带生成龛,以启动肌腱再生的韧带生成。总之,我们认识到NF-κB信号是定制促腱生成龛的关键靶点,并建议将干细胞和TPCA-1的联合递送作为治疗急性肌腱损伤的潜在策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。
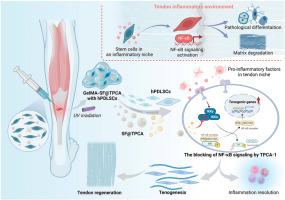
Controlled TPCA-1 delivery engineers a pro-tenogenic niche to initiate tendon regeneration by targeting IKKβ/NF-κB signaling
Tendon repair remains challenging due to its poor intrinsic healing capacity, and stem cell therapy has emerged as a promising strategy to promote tendon regeneration. Nevertheless, the inflammatory environment following acute tendon injuries disrupts stem cell differentiation, leading to unsatisfied outcomes. Our study recognized the critical role of NF-κB signaling in activating inflammation and suppressing tenogenic differentiation of stem cells after acute tendon injury via multiomics analysis. TPCA-1, a selective inhibitor of IKKβ/NF-κB signaling, efficiently restored the impaired tenogenesis of stem cells in the inflammatory environment. By developing a microsphere-incorporated hydrogel system for stem cell delivery and controlled release of TPCA-1, we successfully engineered a pro-tenogenic niche to initiate tenogenesis for tendon regeneration. Collectively, we recognize NF-κB signaling as a critical target to tailor a pro-tenogenic niche and propose the combined delivery of stem cells and TPCA-1 as a potential strategy for acute tendon injuries.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Bioactive Materials
Biochemistry, Genetics and Molecular Biology-Biotechnology
CiteScore
28.00
自引率
6.30%
发文量
436
审稿时长
20 days
期刊介绍:
Bioactive Materials is a peer-reviewed research publication that focuses on advancements in bioactive materials. The journal accepts research papers, reviews, and rapid communications in the field of next-generation biomaterials that interact with cells, tissues, and organs in various living organisms.
The primary goal of Bioactive Materials is to promote the science and engineering of biomaterials that exhibit adaptiveness to the biological environment. These materials are specifically designed to stimulate or direct appropriate cell and tissue responses or regulate interactions with microorganisms.
The journal covers a wide range of bioactive materials, including those that are engineered or designed in terms of their physical form (e.g. particulate, fiber), topology (e.g. porosity, surface roughness), or dimensions (ranging from macro to nano-scales). Contributions are sought from the following categories of bioactive materials:
Bioactive metals and alloys
Bioactive inorganics: ceramics, glasses, and carbon-based materials
Bioactive polymers and gels
Bioactive materials derived from natural sources
Bioactive composites
These materials find applications in human and veterinary medicine, such as implants, tissue engineering scaffolds, cell/drug/gene carriers, as well as imaging and sensing devices.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


