利用偏高岭土 K 基土工聚合物开发用于去除和回收城市污水中氨的离子交换工艺。
IF 8.1
2区 环境科学与生态学
Q1 ENVIRONMENTAL SCIENCES
引用次数: 0
摘要
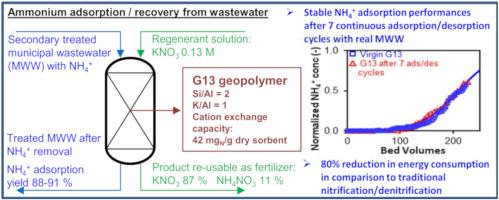
离子交换是从城市污水(MWW)中去除铵,以便将其回收用于肥料生产的一种前景广阔的工艺。以往关于铵离子交换的研究忽视了对工艺稳健性的评估和解吸/回收步骤的优化。本研究旨在开发一种基于偏高岭土 K 基土工聚合物(名为 G13)的氨去除/回收连续流工艺。通过使用两种类型的 MWW 进行 7 个吸附/解吸循环,对工艺的稳健性进行了评估。这些测试的结果令人满意,而且性能稳定:入口处 40 mgN L-1 的运行能力 = 12 mgN gdry 吸附剂-1,选定断点处处理过的 MWW 的床体积 = 199 - 226,铵吸附率 = 88 - 91%。空床接触时间(EBCT)从 10 分钟减少到 5 分钟,但性能没有降低。托马斯模型有效地模拟了 NH4+ 的吸附过程,从而可以基于模型评估减少 EBCT 对工艺性能的影响。创新的解吸程序带来了很高的铵回收率(86 - 100%),解吸产物主要由 KNO3(54%w)和 NH4NO3(39%w)组成,这两种盐在商业化肥中使用量很大。使用 G13 去除/回收氨的能耗为 0.027 kWh m-3(处理过的 WW),与传统的硝化/反硝化工艺相比,能耗明显降低,而运行成本则相当于基准工艺成本的 60-110%。这些结果表明,在循环经济方法中,G13 是一种很有前景的氨回收材料。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Development of an ion exchange process for ammonium removal and recovery from municipal wastewater using a metakaolin K-based geopolymer
Ion exchange represents a promising process for ammonium removal from municipal wastewater (MWW), in order to recover it for fertilizer production. Previous studies on ammonium ion exchange neglected the assessment of process robustness and the optimization the desorption/recovery step. This study aimed at developing a continuous-flow process of ammonium removal/recovery based on a metakaolin K-based geopolymer, named G13. Process robustness was assessed by operating 7 adsorption/desorption cycles with two types of MWW. These tests resulted in satisfactory and constant performances: operating capacity at 40 mgN L−1 in the inlet = 12 mgN gdry sorbent−1, bed volumes of treated MWW at the selected breakpoint = 199–226, ammonium adsorption yield = 88–91%. Empty bed contact time (EBCT) was decreased from 10 to 5 min without any reduction in performances. The NH4+ adsorption process was effectively simulated by the Thomas model, allowing a model-based assessment of the effect of EBCT reductions on process performances. An innovative desorption procedure led to high ammonium recovery yields (86–100%) and to a desorbed product composed primarily of KNO3 (54%w) and NH4NO3 (39%w), two salts largely used in commercial fertilizers. The energy consumption of ammonium removal/recovery with G13 resulted 0.027 kWh m−3treated WW, with a relevant reduction in comparison to traditional nitrification/denitrification, whereas the operational cost resulted equal to 60–110% of the cost of the benchmark process. These results show that G13 is a promising material to recover ammonium in a circular economy approach.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Chemosphere
环境科学-环境科学
CiteScore
15.80
自引率
8.00%
发文量
4975
审稿时长
3.4 months
期刊介绍:
Chemosphere, being an international multidisciplinary journal, is dedicated to publishing original communications and review articles on chemicals in the environment. The scope covers a wide range of topics, including the identification, quantification, behavior, fate, toxicology, treatment, and remediation of chemicals in the bio-, hydro-, litho-, and atmosphere, ensuring the broad dissemination of research in this field.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


