原位形成外环乙烯基烯烃的实用模块化环化反应
IF 11.5
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
活性中间体可以通过化学和区域选择性环化反应快速构建融合和/或桥接环系统,对活性中间体的探索继续引起合成界的极大兴趣。作为 3 碳构建模块的乙烯基羰基化合物经常被用于通过连续形成多个碳-碳和/或碳-异原子键来构建(杂)环框架。在此,我们报告了一种通过选择性金(I)促进叠氮-烯炔环化过程催化生成外环α-乙烯基碳金物种的简明策略。随后,这些原位形成的中间体与不同类型的伙伴进行了实用的模块化环化反应,以高化学、区域和立体选择性生成了一系列不同的融合和桥接吡咯。本文章由计算机程序翻译,如有差异,请以英文原文为准。
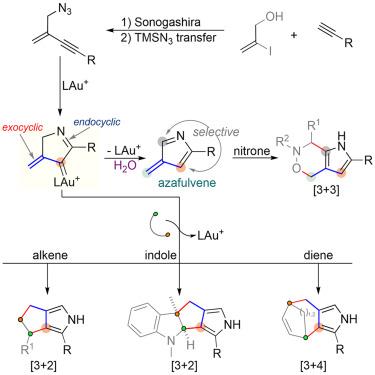
Practical and modular cycloadditions of in-situ formed exocyclic vinylcarbenes
The exploration of reactive intermediates, which enable chemo- and regioselective cycloaddition reactions for the expeditious construction of fused and/or bridged ring systems, continues to draw a great deal of interest from the synthetic community. Vinylcarbene species, which serve as 3-carbon building blocks, have been frequently used for the construction of (hetero)cyclic frameworks through the successive formation of multiple carbon–carbon and/or carbon–heteroatom bonds. Herein, we report a concise strategy for the catalytic generation of an exocyclic α-vinyl gold carbene species via a selective gold(I)-promoted azide-enyne cyclization process. Subsequently, practical and modular cycloadditions of these in-situ-formed intermediates with different types of partners were disclosed, producing a diverse array of fused and bridged pyrroles in high chemo-, regio-, and stereoselectivity.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
10.50
自引率
6.40%
发文量
0
期刊介绍:
Chem Catalysis is a monthly journal that publishes innovative research on fundamental and applied catalysis, providing a platform for researchers across chemistry, chemical engineering, and related fields. It serves as a premier resource for scientists and engineers in academia and industry, covering heterogeneous, homogeneous, and biocatalysis. Emphasizing transformative methods and technologies, the journal aims to advance understanding, introduce novel catalysts, and connect fundamental insights to real-world applications for societal benefit.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


