脑血管和脑实质之间的硬度差异通过血管共用促进转移浸润
IF 17.3
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
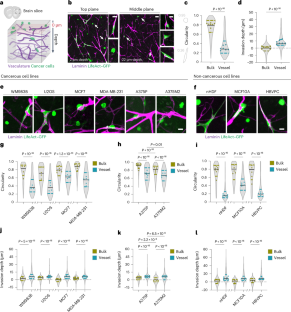
在脑转移过程中,癌细胞与现有血管保持密切接触,并可利用血管作为迁移路径--这一过程被称为血管共用。然而,人们对这种迁移形式的调控机制知之甚少。在这里,我们利用体外脑切片和体外血管共择有机模型,证明癌细胞沿脑血管入侵是由血管和脑实质之间的硬度差异驱动的。成像分析表明,细胞通过粘附在基底膜细胞外基质上沿血管基底面移动。我们进一步发现,脑血管的硬度和周围环境的柔软度都会增强血管的粘附性,前者通过滑石蛋白依赖机制加强病灶粘附,后者则允许细胞移动。我们的研究揭示了一种机械传感机制,它能引导细胞迁移以应对组织内在的机械异质性,这对癌症的侵袭和转移具有重要意义。本文章由计算机程序翻译,如有差异,请以英文原文为准。

Differential stiffness between brain vasculature and parenchyma promotes metastatic infiltration through vessel co-option
In brain metastasis, cancer cells remain in close contact with the existing vasculature and can use vessels as migratory paths—a process known as vessel co-option. However, the mechanisms regulating this form of migration are poorly understood. Here we use ex vivo brain slices and an organotypic in vitro model for vessel co-option to show that cancer cell invasion along brain vasculature is driven by the difference in stiffness between vessels and the brain parenchyma. Imaging analysis indicated that cells move along the basal surface of vessels by adhering to the basement membrane extracellular matrix. We further show that vessel co-option is enhanced by both the stiffness of brain vasculature, which reinforces focal adhesions through a talin-dependent mechanism, and the softness of the surrounding environment that permits cellular movement. Our work reveals a mechanosensing mechanism that guides cell migration in response to the tissue’s intrinsic mechanical heterogeneity, with implications in cancer invasion and metastasis. Uroz et al. report that the distinct mechanical properties of brain vasculature versus parenchyma drive cancer cell migration through a talin-dependent mechanism, enabling vessel co-option and metastatic invasion in the brain.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Nature Cell Biology
生物-细胞生物学
CiteScore
28.40
自引率
0.90%
发文量
219
审稿时长
3 months
期刊介绍:
Nature Cell Biology, a prestigious journal, upholds a commitment to publishing papers of the highest quality across all areas of cell biology, with a particular focus on elucidating mechanisms underlying fundamental cell biological processes. The journal's broad scope encompasses various areas of interest, including but not limited to:
-Autophagy
-Cancer biology
-Cell adhesion and migration
-Cell cycle and growth
-Cell death
-Chromatin and epigenetics
-Cytoskeletal dynamics
-Developmental biology
-DNA replication and repair
-Mechanisms of human disease
-Mechanobiology
-Membrane traffic and dynamics
-Metabolism
-Nuclear organization and dynamics
-Organelle biology
-Proteolysis and quality control
-RNA biology
-Signal transduction
-Stem cell biology
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


