用于超强、持久抗病毒治疗的自适应多表位靶向和抗体增强型纳米抗体平台
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
病原体会不断进化,并发生突变,从而逃避宿主的免疫和治疗。解决这些逃逸机制需要靶向进化上保守的薄弱环节,因为这些区域的突变往往会带来健康代价。我们介绍了一种模块化多价纳米抗体平台--增强型自适应多表位靶向(AMETA),它将强效双特异性纳米抗体与人类免疫球蛋白M(IgM)支架连接在一起。AMETA 可以显示 20 多种纳米抗体,从而实现与多个保守表位和中和表位的超强结合。通过利用多表位 SARS-CoV-2 纳米抗体和结构引导设计,AMETA 构建物成倍提高了抗病毒效力,超过单体纳米抗体一百万倍以上。这些构建体对致病性沙巴病毒(包括 Omicron 亚系)具有超强、广泛和持久的疗效,并取得了可靠的临床前结果。通过冷冻电子显微镜和建模进行的结构分析发现了单个构建体的多种抗病毒机制。在皮摩尔至纳摩尔浓度下,AMETA 能有效诱导尖峰间和病毒间的交联,促进尖峰后融合并显著解除病毒。AMETA 的模块化设计可实现快速、经济高效的生产,并适应不断变化的病原体。本文章由计算机程序翻译,如有差异,请以英文原文为准。
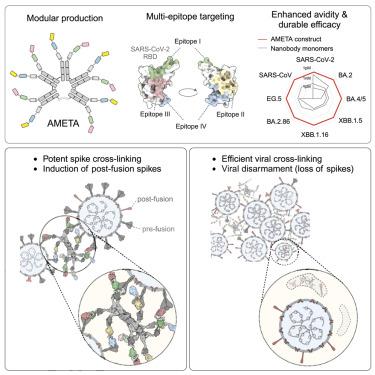
Adaptive multi-epitope targeting and avidity-enhanced nanobody platform for ultrapotent, durable antiviral therapy
Pathogens constantly evolve and can develop mutations that evade host immunity and treatment. Addressing these escape mechanisms requires targeting evolutionarily conserved vulnerabilities, as mutations in these regions often impose fitness costs. We introduce adaptive multi-epitope targeting with enhanced avidity (AMETA), a modular and multivalent nanobody platform that conjugates potent bispecific nanobodies to a human immunoglobulin M (IgM) scaffold. AMETA can display 20+ nanobodies, enabling superior avidity binding to multiple conserved and neutralizing epitopes. By leveraging multi-epitope SARS-CoV-2 nanobodies and structure-guided design, AMETA constructs exponentially enhance antiviral potency, surpassing monomeric nanobodies by over a million-fold. These constructs demonstrate ultrapotent, broad, and durable efficacy against pathogenic sarbecoviruses, including Omicron sublineages, with robust preclinical results. Structural analysis through cryoelectron microscopy and modeling has uncovered multiple antiviral mechanisms within a single construct. At picomolar to nanomolar concentrations, AMETA efficiently induces inter-spike and inter-virus cross-linking, promoting spike post-fusion and striking viral disarmament. AMETA’s modularity enables rapid, cost-effective production and adaptation to evolving pathogens.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


