利用测得的元素组成模拟碳质介相热力学
IF 3.8
3区 工程技术
Q2 ENGINEERING, CHEMICAL
引用次数: 0
摘要
本文章由计算机程序翻译,如有差异,请以英文原文为准。
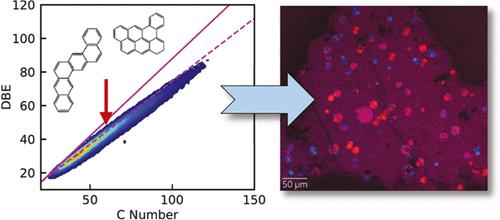
Modeling Carbonaceous Mesophase Thermodynamics Using Measured Elemental Compositions
Carbonaceous mesophase is a liquid crystalline material that can form as petroleum is pyrolyzed into coke. The material is thought to be made up of discotic mesogens in the form of planar, polyaromatic molecules. Researchers have previously developed thermodynamic models to predict mesophase formation in different feeds based on molecular weight, independent of the chemical structures found in any given sample of material. Using Fourier transform ion cyclotron resonance mass spectroscopy (FT-ICR MS), we were able to observe how the elemental compositions of hydrocarbon molecules evolved with thermal treatment. Using this information, we devised a new approach for modeling petroleum-based liquid crystal phase behavior using both molecular weight and elemental compositions to describe the material’s composition. This approach permits molecules of large molecular weight to not automatically be considered mesogens and for nonideal solution behavior and mesophase content to be parametrized independently. Through comparing different methods, we also demonstrated the important role nonideal solution effects have on the calculated phase behavior as well as the impact of pseudocomponent number on the size of the mesophase coexistence region. Further theoretical and experimental work is needed to develop more general, predictive models.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Industrial & Engineering Chemistry Research
工程技术-工程:化工
CiteScore
7.40
自引率
7.10%
发文量
1467
审稿时长
2.8 months
期刊介绍:
ndustrial & Engineering Chemistry, with variations in title and format, has been published since 1909 by the American Chemical Society. Industrial & Engineering Chemistry Research is a weekly publication that reports industrial and academic research in the broad fields of applied chemistry and chemical engineering with special focus on fundamentals, processes, and products.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


