体外释放测试(IVRT)的合成膜选择:糠酸莫米松局部半固体制剂的案例研究。
IF 4.3
3区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
摘要
体外释放试验(IVRT)被广泛用于开发外用半固体产品的配方,评估产品在放大和批准后变更后的质量和一致性,最近还被用于帮助评估外用非专利产品的等效性。合成膜的选择是 IVRT 方法开发过程中最关键的参数之一。众所周知,膜的特性,即其聚合物基质、孔隙率、孔径、厚度会对 IVRT 数据产生重大影响。然而,文献中并没有关于不同类型外用剂型膜选择的详细信息。因此,我们旨在研究相同药物分子的不同外用半固体剂型是否会导致膜选择的变化。在此框架内,我们主要研究了市售的三种不同类型的糠酸莫米松(0.1%)外用半固体制剂(软膏、乳膏和洗剂)的流变特性。然后,在确定了确保糠酸莫米松沉降条件的适当受体介质后,使用一系列合成亲水膜(再生纤维素、醋酸纤维素、硝酸纤维素、混合纤维素酯膜)和疏水膜(聚醚砜、聚丙烯、聚四氟乙烯膜)进行了膜惰性试验。最后,利用弗朗兹型扩散池对外用半固体产品进行了 IVRT 研究。膜的惰性和体外释放数据表明,醋酸纤维素膜总体上表现出较好的扩散特性,而其他合成膜在糠酸莫米松的不同半固体剂型上表现出不同的结果。总之,研究结果表明,不同的合成膜在 6 小时后的药物释放率和累积释放量可能因半固体剂型而异。为了选择用于 IVRT 的合成膜,在进行膜惰性试验和 IVRT 研究之前,还应考虑聚合物基质与药物分子的化学结构以及制剂成分之间的潜在相互作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
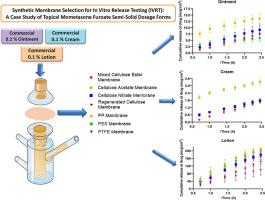
Synthetic membrane selection for in vitro release testing (IVRT): A case study of topical mometasone furoate semi-solid dosage forms
In vitro release testing (IVRT) is extensively used to develop the formulation of topical semi-solid products, to evaluate the quality and consistency of the product after scale-up and post-approval changes, and more recently to aid the evaluation of topical generic products’ equivalency. The selection of synthetic membrane is one of the most critical parameters of the method development part of IVRT. It is well known that the membrane features namely its polymer matrices, porosity, pore size, thickness can have a substantial effect on the IVRT data. However, there is no detailed information available in the literature regarding the membrane selection for different types of topical dosage forms. Therefore, we aimed to investigate whether difference topical semi-solid dosage forms of the same drug molecule would cause to variation in the membrane selection. Within this framework, rheological behaviour of commercially available three different types of topical semi-solid dosage forms (ointment, cream, and lotion) of mometasone furoate (0.1%) were primarily characterized. Then, the membrane inertness test was conducted using a series of synthetic hydrophilic membranes (regenerated cellulose, cellulose acetate, cellulose nitrate, mixed cellulose ester membranes) and hydrophobic membranes (polyether sulfone, polypropylene, polytetrafluoroethylene membranes) after identifying of an appropriate receptor medium that ensures sink condition for mometasone furoate. Lastly, IVRT studies from the topical semi-solid products were performed utilizing Franz-type diffusion cells. The membrane inertness and in vitro release data demonstrated that the cellulose acetate membrane showed superior diffusion properties in general while the other synthetic membranes exhibited varying outcomes for different semi-solid dosage forms of mometasone furoate. Overall, the results indicated that the release rate and the cumulative drug released amount of drug after 6 h through the different synthetic membranes might vary depending on the semi-solid dosage form. In order to select the synthetic membrane for IVRT, it should also be considered potential interactions between the polymer matrices and the chemical structure of drug molecule as well as formulation components prior to conducting membrane inertness test and IVRT studies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
9.60
自引率
2.20%
发文量
248
审稿时长
50 days
期刊介绍:
The journal publishes research articles, review articles and scientific commentaries on all aspects of the pharmaceutical sciences with emphasis on conceptual novelty and scientific quality. The Editors welcome articles in this multidisciplinary field, with a focus on topics relevant for drug discovery and development.
More specifically, the Journal publishes reports on medicinal chemistry, pharmacology, drug absorption and metabolism, pharmacokinetics and pharmacodynamics, pharmaceutical and biomedical analysis, drug delivery (including gene delivery), drug targeting, pharmaceutical technology, pharmaceutical biotechnology and clinical drug evaluation. The journal will typically not give priority to manuscripts focusing primarily on organic synthesis, natural products, adaptation of analytical approaches, or discussions pertaining to drug policy making.
Scientific commentaries and review articles are generally by invitation only or by consent of the Editors. Proceedings of scientific meetings may be published as special issues or supplements to the Journal.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


