通过固态核磁共振波谱和分子动力学模拟研究天冬氨酸与形态各异的羟基磷灰石纳米颗粒的结合情况
IF 3.7
2区 化学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
羟基磷灰石(HAP)在生物硬组织中呈现出高度定向的分层结构。HAP 的形成和选择性结晶取向是一个涉及富含酸性残基的功能性生物矿化蛋白的过程。为了深入了解 HAP 矿化和酸性残基结合的过程,在吸附天冬氨酸(Asp)后,对合成的具有特定晶格面(包括 (001)、(100) 和 (011) )的 HAP 进行了结构表征。对天冬氨酸在 HAP 表面的吸附亲和力进行了定量评估,结果表明吸附亲和力与 HAP 形态有很大关系。在合成的 HAP 纳米粒子(NPs)中,Asp 对由 (100) 和 (011) 晶格面组成的短 HAP 纳米棒的吸附亲和力最强,其次是优先表现为 (001) 面的纳米片,Asp 对其的吸附亲和力相似但略低。优先呈现 (100) 晶格面的 HAP 纳米线与前两种 HAP 纳米粒子相比,对 Asp 的亲和力明显降低,并有多层吸附的迹象。该研究结合了固态核磁共振(SSNMR)技术,包括 13C 和 15N CP-MAS、弛豫测量和 13C-31P 旋转回波可溶共振(REDOR),以表征富含 13C 和 15N Asp 的 Asp-HAP 双纳米界面的分子结构和动力学。REDOR 用于确定 13C-31P 核间距离,从而深入了解 Asp 结合的几何形状,在这种几何形状中,较强的 13C-31P 偶极耦合与根据朗缪尔等温线确定的结合亲和力相关。羧基位点被确定为主要的结合基团,它们与表面钙位点的相互作用促进了这种结合。通过分子动力学(MD)模拟进一步完善了 SSNMR 揭示的 Asp 螯合构象,SSNMR 和 MD 模型对不同表面的特定模型非常吻合。本文章由计算机程序翻译,如有差异,请以英文原文为准。
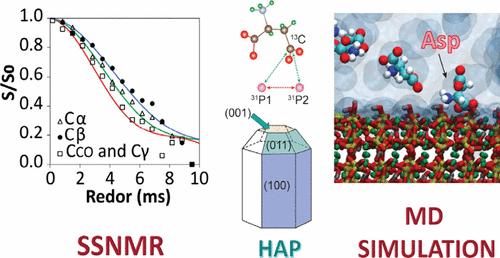
Aspartic Acid Binding on Hydroxyapatite Nanoparticles with Varying Morphologies Investigated by Solid-State NMR Spectroscopy and Molecular Dynamics Simulation
Hydroxyapatite (HAP) exhibits a highly oriented hierarchical structure in biological hard tissues. The formation and selective crystalline orientation of HAP is a process that involves functional biomineralization proteins abundant in acidic residues. To obtain insights into the process of HAP mineralization and acidic residue binding, synthesized HAP with specific lattice planes including (001), (100), and (011) are structurally characterized following the adsorption of aspartic acid (Asp). The adsorption affinity of Asp on HAP surfaces is evaluated quantitatively and demonstrates a high dependency on the HAP morphological form. Among the synthesized HAP nanoparticles (NPs), Asp exhibits the strongest adsorption affinity to short HAP nanorods, which are composed of (100) and (011) lattice planes, followed by nanosheets with a preferential expression of the (001) facet, to which Asp displays a similar but slightly lower binding affinity. HAP nanowires, with the (100) lattice plane preferentially developed, show significantly lower affinity to Asp and evidence of multilayer adsorption compared to the previous two types of HAP NPs. A combination of solid-state NMR (SSNMR) techniques including 13C and 15N CP-MAS, relaxation measurements and 13C–31P Rotational Echo DOuble Resonance (REDOR) is utilized to characterize the molecular structure and dynamics of Asp-HAP bionano interfaces with 13C- and 15N-enriched Asp. REDOR is used to determine 13C–31P internuclear distances, providing insight into the Asp binding geometry where stronger 13C–31P dipolar couplings correlate with binding affinity determined from Langmuir isotherms. The carboxyl sites are identified as the primary binding groups, facilitated by their interaction with surface calcium sites. The Asp chelation conformations revealed by SSNMR are further refined with molecular dynamics (MD) simulation where specific models strongly agree between the SSNMR and MD models for the various surfaces.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Langmuir
化学-材料科学:综合
CiteScore
6.50
自引率
10.30%
发文量
1464
审稿时长
2.1 months
期刊介绍:
Langmuir is an interdisciplinary journal publishing articles in the following subject categories:
Colloids: surfactants and self-assembly, dispersions, emulsions, foams
Interfaces: adsorption, reactions, films, forces
Biological Interfaces: biocolloids, biomolecular and biomimetic materials
Materials: nano- and mesostructured materials, polymers, gels, liquid crystals
Electrochemistry: interfacial charge transfer, charge transport, electrocatalysis, electrokinetic phenomena, bioelectrochemistry
Devices and Applications: sensors, fluidics, patterning, catalysis, photonic crystals
However, when high-impact, original work is submitted that does not fit within the above categories, decisions to accept or decline such papers will be based on one criteria: What Would Irving Do?
Langmuir ranks #2 in citations out of 136 journals in the category of Physical Chemistry with 113,157 total citations. The journal received an Impact Factor of 4.384*.
This journal is also indexed in the categories of Materials Science (ranked #1) and Multidisciplinary Chemistry (ranked #5).
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


