通过设计具有核壳结构域的 Arc 蛋白来增强 mRNA 的相互作用
IF 3.7
2区 化学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
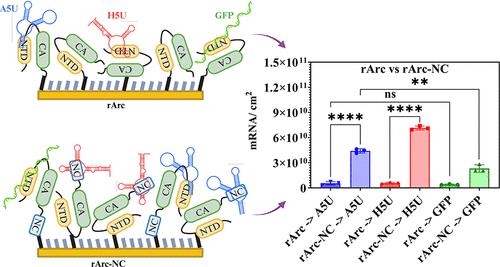
活性调节细胞骨架相关蛋白(Arc)形成类似病毒的囊壳,用于在神经元之间运输 mRNA。哺乳动物 Arc 与 HIV-1 群体特异性抗原(Gag)不同,后者使用其核噬菌体(NC)结构域结合 HIV-1 基因组 mRNA,而 Arc 则缺乏 NC 结构域,因此它们直接结合 mRNA 的相互作用仍未得到充分探索。本研究检测了大鼠 Arc 与大鼠 Arc 5′ UTR (A5U)、HIV-1 5′ UTR (H5U) 和 GFP mRNA 的结合情况,结果显示它们的结合很弱,没有明显的偏好。在 rArc 的 C 端添加 HIV-1 NC 结构域后,与 H5U 的结合力显著提高,同时与 A5U 的结合力也达到了其 H5U 水平的 60%,与 A5U 的亲和力是 GFP mRNA 的两倍。重要的是,rArc-NC 结合的 A5U 和 H5U 是 GST-NC 的 3.4 倍,这表明 rArc NTD-CA 有助于 HIV-1 NC 结合 mRNA。这些发现表明了一种保守的 Gag 蛋白-mRNA 相互作用机制,凸显了将内源性 Gag NTD-CA 与逆转录病毒 NC 和 UTR 结合起来开发 mRNA 运送系统的潜力。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Enhancing mRNA Interactions by Engineering the Arc Protein with Nucleocapsid Domain
Activity-regulated cytoskeleton-associated protein (Arc) forms virus-like capsids for mRNA transport between neurons. Unlike HIV-1 Group-specific Antigen (Gag), which uses its Nucleocapsid (NC) domain to bind HIV-1 genomic mRNA, mammalian Arc lacks the NC domain, and their direct mRNA binding interactions remain underexplored. This study examined rat Arc’s binding to rat Arc 5′ UTR (A5U), HIV-1 5′ UTR (H5U), and GFP mRNAs, revealing weak binding with no significant preference. Adding the HIV-1 NC domain to rArc’s C-terminus significantly improved binding to H5U, while also showing substantial binding to A5U at about 60% of its H5U level and exhibiting twice the affinity for A5U over GFP mRNA. Importantly, rArc-NC binds 3.4 times more A5U and H5U than GST-NC, indicating that rArc NTD-CA aids mRNA binding by HIV-1 NC. These findings suggest a conserved Gag protein–mRNA interaction mechanism, highlighting the potential for developing mRNA delivery systems that combine endogenous Gag NTD-CA with retroviral NC and UTRs.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Langmuir
化学-材料科学:综合
CiteScore
6.50
自引率
10.30%
发文量
1464
审稿时长
2.1 months
期刊介绍:
Langmuir is an interdisciplinary journal publishing articles in the following subject categories:
Colloids: surfactants and self-assembly, dispersions, emulsions, foams
Interfaces: adsorption, reactions, films, forces
Biological Interfaces: biocolloids, biomolecular and biomimetic materials
Materials: nano- and mesostructured materials, polymers, gels, liquid crystals
Electrochemistry: interfacial charge transfer, charge transport, electrocatalysis, electrokinetic phenomena, bioelectrochemistry
Devices and Applications: sensors, fluidics, patterning, catalysis, photonic crystals
However, when high-impact, original work is submitted that does not fit within the above categories, decisions to accept or decline such papers will be based on one criteria: What Would Irving Do?
Langmuir ranks #2 in citations out of 136 journals in the category of Physical Chemistry with 113,157 total citations. The journal received an Impact Factor of 4.384*.
This journal is also indexed in the categories of Materials Science (ranked #1) and Multidisciplinary Chemistry (ranked #5).
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


