杂环 N1-N2 键裂解过程中产生的骨架重组:电合成方法
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
我们设计了一种通过重组分子骨架合成功能化二氮杂卓和喹唑啉的统一方法。该方法以噌啉类和吲唑类为设计前体,通过多米诺 N1-N2 键裂解和同时闭环,利用电能进行合成。此外,还通过分子间的环同源反应,以 2,3-二芳基吲唑和乙腈为原料,采用相同的电化学策略合成了致密官能化的二氢喹唑啉。本文章由计算机程序翻译,如有差异,请以英文原文为准。
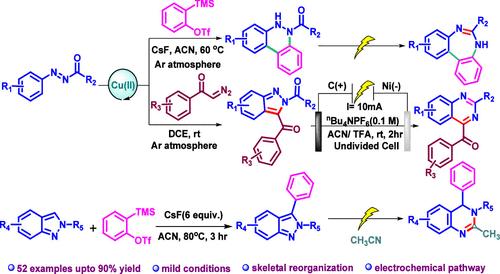
Skeletal Reorganization Emanated via the Course of Heterocyclic N1–N2 Bond Cleavage: Electrosynthetic Approach
A unified method toward the synthesis of functionalized diazepines and quinazolines through reorganization of the molecular skeleton has been devised. The process is indulged by electrical energy via a domino N1–N2 bond cleavage followed by concomitant ring closing, initiating from cinnolines and indazoles as designed precursors. Additionally, an intermolecular ring homologation has also been established to synthesize densely functionalized dihydroquinazolines from 2,3-diaryl-indazoles and acetonitrile involving the same electrochemical strategy.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


