调节膦酰胺的电子结构以绕过膦进行催化阿贝尔反应
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
我们制备了一组结构类似但电子结构不同的膦酰胺(1aa-10aa),并测试了对取代导致的电子修正对通过阿贝尔反应将醇类转化为卤化物的影响。OMe-取代的二磷酰胺(8aa)仍然是最活跃的,在底物范围较大的中等反应条件下,可将醇类转化为卤化物的转化率高达 ∼96%,吨转化率为 11。卤化物的形成遵循假一阶速率,恒定速率(kobs)为 7.13 × 10-5 s-1。温度相关动力学和艾林分析显示,活化参数ΔH‡为 28.95 (±1.6) kcal mol-1,ΔS‡为 -70.02 (±0.4) cal K-1 mol-1,ΔG‡298 为 49.81 (±1.2) kcal mol-1。氘标记研究表明,醇的 O-H 解离是决定速率的步骤,而对取代苄醇的 Hammett 分析表明,在阿贝尔反应过程中,磷中心积累了正电荷。HOMO-LUMO 能量和 NPA 分析表明,8aa 中的 p-OMe 取代使 "P═O "键更具离子性,相应的亲氨基膦具有亲核性,这些都有利于阿贝尔反应。原位检测阿贝尔盐 [R3PX]CX3 和烷氧基鏻阳离子 [R3POR]X 验证了由膦酰胺介导的反应途径。本文章由计算机程序翻译,如有差异,请以英文原文为准。
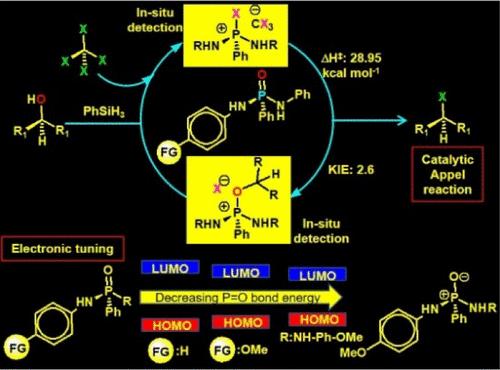
Moderation of the Electronic Structure of Phosphamides to Execute the Catalytic Appel Reaction Bypassing Phosphine
A set of structurally analogous, albeit electronically distinct, phosphamides (1aa-10aa) is prepared, and the effect of the electronic amendment due to p-substitution has been tested for the conversion of alcohols to halides via the Appel reaction. The −OMe-substituted diphosphamide (8aa) remains the most active, providing ∼96% conversion of alcohols to halides with a TON of 11 in moderate reaction conditions with a large substrate scope. Halide formation follows a pseudo-first-order rate with a constant rate (kobs) of 7.13 × 10–5 s–1. Temp-dependent kinetics and Eyring analyses reveal the activation parameters ΔH‡ of 28.95 (±1.6) kcal mol–1, ΔS‡ of −70.02 (±0.4) cal K–1 mol–1, and ΔG‡298 of 49.81 (±1.2) kcal mol–1. The deuterium labeling study highlights the O–H dissociation of the alcohol as the rate-determining step, while the Hammett analysis with p-substituted benzyl alcohols indicates a positive charge accumulation at the phosphorus center during the Appel reaction. The HOMO–LUMO energy and NPA analyses show that p–OMe substitutions in 8aa make the “P═O” bond more ionic and corresponding aminophosphine is nucleophilic, which are favorable for the Appel reaction. In situ detection of the Appel salt, [R3PX]CX3 and alkoxy phosphonium cation [R3POR]X, validates the reaction pathway mediated by the phosphamides.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


