基于细胞的筛选发现了一种用于治疗多药耐药性的强效口服 ABCB1 调节剂
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
以 ABCB1 为靶点是一种很有前景的抗多药策略。通过基于细胞的表型筛选,我们发现了新型三唑并[1,5-a]嘧啶酮类 ABCB1 调节剂。值得注意的是,WS-917 被确定为提高人结直肠腺癌细胞(SW620/Ad300)对紫杉醇敏感性(IC50 = 5 nM)的重要因素。机理分析表明,该化合物大大增加了细胞内紫杉醇和[3H]-紫杉醇的浓度,同时通过抑制 ABCB1 的外流减轻了 SW620/Ad300 中[3H]-紫杉醇的外流。细胞热转移试验强调了 WS-917 通过直接结合稳定 ABCB1 的能力。此外,WS-917 还能刺激 ABCB1 ATPase 的活性,同时对 CYP3A4 的抑制作用微乎其微。值得注意的是,WS-917 还能提高 SW620/Ad300 对紫杉醇的敏感性,以及 CT26/TAXOL 对紫杉醇和 PD-L1 抑制剂(Atezolizumab)的敏感性,所有这些都不会引起明显的毒性。WS-917 的发现为开发更有效的 ABCB1 调节剂带来了希望。本文章由计算机程序翻译,如有差异,请以英文原文为准。
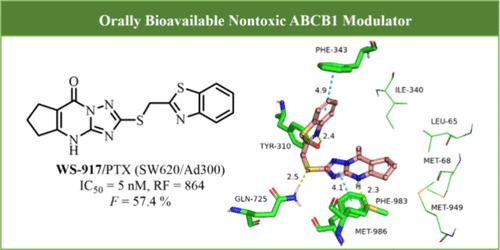
Cell-Based Screen Identifies a Highly Potent and Orally Available ABCB1 Modulator for Treatment of Multidrug Resistance
Targeting ABCB1 is a promising strategy in combating multidrug resistance. Our cell-based phenotypic screening led to the discovery of novel triazolo[1,5-a]pyrimidone-based ABCB1 modulators. Notably, WS-917 was identified as a significant contributor to heightened sensitization of human colorectal adenocarcinoma cells (SW620/Ad300) to paclitaxel (IC50 = 5 nM). Mechanistic elucidation revealed that this compound substantially augmented intracellular paclitaxel and [3H]-paclitaxel, concurrently mitigating the efflux of [3H]-paclitaxel in SW620/Ad300 through the inhibition of ABCB1 efflux. The cellular thermal shift assay underscored its ability to stabilize ABCB1 through direct binding. Additionally, WS-917 induced stimulation of ABCB1 ATPase activity while exhibiting negligible inhibitory effect against CYP3A4. Remarkable was its capacity to enhance the sensitivity of SW620/Ad300 to paclitaxel, as well as the sensitivity of CT26/TAXOL to paclitaxel and PD-L1 inhibitor (Atezolizumab) in vivo, all achieved without inducing observable toxicity. The discovery of WS-917 holds promise for the development of more potent ABCB1 modulators.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


