福莫西汀通过依赖肠道微生物群的方式重塑 M1/M2 巨噬细胞极化的平衡来缓解溃疡性结肠炎
IF 6.7
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
背景溃疡性结肠炎(UC)是炎症性肠病的一种,由于现有药物的局限性,临床治疗面临巨大挑战。本研究旨在探讨 FN 对 UC 的疗效及其潜在的调节机制。方法采用葡聚糖硫酸钠(DSS)复制小鼠实验性结肠炎,并同时进行 FN 治疗。用免疫荧光观察结肠组织中 CD68 和 F4/80 巨噬细胞的分布和定位,用 ELISA 测定其趋化因子和炎性细胞因子的浓度,用流式细胞术测定巨噬细胞和 M1/M2 亚群。此外,16 s rRNA 和 LC-MS 技术分别用于检测结肠肠道微生物群和代谢物谱。结果FN剂量依赖性地减轻了结肠炎小鼠的临床症状和结肠组织的炎症损伤,其大剂量疗效与5-ASA相当。同时,FN 不仅能抑制结肠炎小鼠巨噬细胞的炎性浸润及其 M1/M2 极化平衡,还能改善结肠微生物群的组成和代谢物谱。然而,FN 对 DSS 诱导的结肠炎失去了保护作用,而且在通过四重抗生素治疗消耗肠道菌群之后,FN 无法恢复 M1/M2 巨噬细胞分化的平衡。重要的是,移植经 FN 处理的小鼠粪便微生物群可恢复 FN 对 DSS 诱导的结肠炎的保护作用,并重建其在 M1/M2 巨噬细胞极化中的调节作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
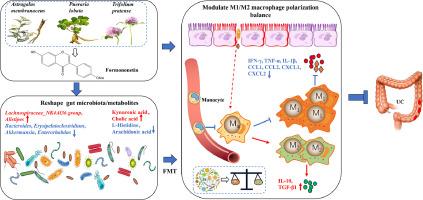
Formononetin alleviates ulcerative colitis via reshaping the balance of M1/M2 macrophage polarization in a gut microbiota-dependent manner
Background
Ulcerative colitis (UC), a type of inflammatory bowel disease, presents substantial challenges in clinical treatment due to the limitations of current medications. Formononetin (FN), a naturally compound with widespread availability, exhibits anti-inflammatory, antioxidant, and immunomodulatory properties.
Purpose
This study aimed to investigate the efficacy of FN against UC and its potential regulatory mechanism.
Methods
Here, dextran sulfate sodium (DSS) was employed to replicate experimental colitis in mice with concomitant FN treatment. The distribution and localisation of CD68 and F4/80 macrophages in colonic tissues were visualized by immunofluorescence, their chemokine and inflammatory cytokine concentrations were determined by ELISA, and macrophages and M1/M2 subpopulations were determined by flow cytometry. Additionally, 16 s rRNA and LC-MS techniques were used to detect the colonic intestinal microbiota and metabolite profiles, respectively. Correlation analyses was performed to clarify the interactions between differential bacteria, metabolites and M1/M2 macrophages, and pseudo sterile mice were constructed by depletion of gut flora with quadruple antibiotics, followed by faecal microbial transplantation to evaluate its effects on colitis and M1/M2 macrophage polarisation.
Results
FN dose-dependently alleviated clinical symptoms and inflammatory injury in colonic tissues of colitis mice, with its high-dose efficacy comparable to that of 5-ASA. Concurrently, FN not only inhibited inflammatory infiltration of macrophages and their M1/M2 polarisation balance in colitis mice, but also improved the composition of colonic microbiota and metabolite profiles. However, FN lost its protective effects against DSS-induced colitis and failed to restore the equilibrium of M1/M2 macrophage differentiation following intestinal flora depletion through quadruple antibiotic treatment. Importantly, fecal microbiota transplantation from FN-treated mice restored FN's protective effects against DSS-induced colitis and reestablished its regulatory role in M1/M2 macrophage polarization.
Conclusion
Collectively, FN ameliorated UC through modulating the balance of M1/M2 macrophage polarization in a gut microbiota-dependent manner.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Phytomedicine
医学-药学
CiteScore
10.30
自引率
5.10%
发文量
670
审稿时长
91 days
期刊介绍:
Phytomedicine is a therapy-oriented journal that publishes innovative studies on the efficacy, safety, quality, and mechanisms of action of specified plant extracts, phytopharmaceuticals, and their isolated constituents. This includes clinical, pharmacological, pharmacokinetic, and toxicological studies of herbal medicinal products, preparations, and purified compounds with defined and consistent quality, ensuring reproducible pharmacological activity. Founded in 1994, Phytomedicine aims to focus and stimulate research in this field and establish internationally accepted scientific standards for pharmacological studies, proof of clinical efficacy, and safety of phytomedicines.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


