用于光催化二氧化碳还原和芳基烯烃氧化耦合系统的 Pt1Bi 单原子合金
IF 13.1
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
太阳能驱动的二氧化碳还原为生产燃料和增值化学品提供了一种前景广阔的策略,但该系统的效率和经济性往往受到耦合氧化半反应的限制。在此,我们开发了一种光催化二氧化碳还原为一氧化碳和芳基烯烃氧化为芳基酮的协同系统,该系统采用 TiOx 支持的 Pt1Bi 单原子合金光催化剂,具有高效率和更好的原子经济性。实验和计算研究相结合,阐明了在黑暗中从 Bi 到 Pt 的自发电子转移,这种转移在光辐射下得到增强。密度泛函理论计算表明,芳基烯烃的氧化过程有利于 CO2 的质子化和 COOH* 向 CO* 的解离,而 Bi 则有利于 CO2 的质子化和 CO 在铂位点上的解吸。以α-甲基苯乙烯为底物,CO 的生成速率达到 391 μmol g-1 h-1,电子选择性为 91.4%;苯乙酮产物的产量为 449 μmol g-1 h-1,选择性为 95.5%。本文章由计算机程序翻译,如有差异,请以英文原文为准。
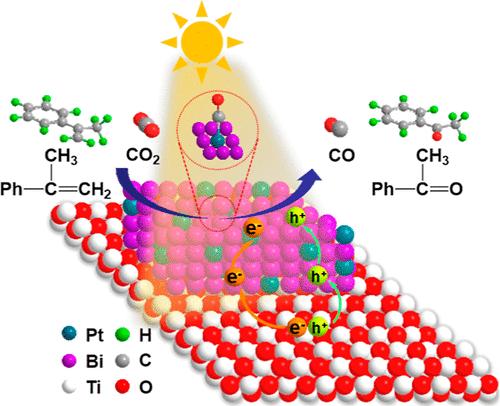
Pt1Bi Single-Atom Alloy for a Coupled Photocatalytic CO2 Reduction and Aryl Olefin Oxidation System
Solar-driven CO2 reduction provides a promising strategy to produce fuels and value-added chemicals, while the efficiency and economy of the system are often limited by the coupled oxidation half-reaction. Here, we develop a synergistic system coupling photocatalytic CO2 reduction to CO and aryl olefin oxidation to aryl ketone with a high-efficiency and improved atom economy over a TiOx-supported Pt1Bi single-atom alloy photocatalyst. Combined experimental and computational studies elucidate the spontaneous electron transfer from Bi to Pt in the dark, which is enhanced under light radiation. Density functional theory calculations reveal that the oxidation process of aryl olefin favors the protonation of CO2 and the dissociation of COOH* to CO*, and Bi facilitates CO2 protonation and CO desorption on the Pt site. The generation rate of CO reaches 391 μmol g–1 h–1 with an electron selectivity of 91.4% using alpha-methylstyrene as the substrate, and the yield of the acetophenone product is 449 μmol g–1 h–1 with a 95.5% selectivity.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


