通过 GPR56 感知类固醇激素 17α-hydroxypregnenolone 可防止铁中毒引起的肝损伤
IF 27.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
G 蛋白偶联受体(GPCR)介导了大多数细胞对激素、神经递质和环境刺激物的反应。然而,GPCR 是否通过铁变态反应参与组织稳态仍不清楚。在这里,我们发现 GPR56/ADGRG1 可使细胞对铁蛋白沉积产生抗性,缺乏 GPR56 会加剧多柔比星(DOX)或缺血再灌注(IR)诱导的铁蛋白沉积介导的肝损伤。从机制上讲,GPR56通过促进CD36的内吞-溶酶体降解,降低了含有游离多不饱和脂肪酸(PUFA)的磷脂的丰度。通过筛选一系列类固醇激素,我们发现 17α-hydroxypregnenolone (17-OH PREG) 可作为 GPR56 的激动剂来拮抗铁蛋白沉积,并有效减轻损伤前后的肝损伤。此外,与疾病相关的 GPR56 突变体对 17-OH PREG 的激活反应迟钝,不足以抵御铁蛋白沉积。总之,我们的研究结果揭示了 17-OH PREG-GPR56 轴介导的信号转导是维持肝脏稳态的一种新的抗铁细胞生成途径,为肝损伤的潜在治疗提供了新的见解。本文章由计算机程序翻译,如有差异,请以英文原文为准。
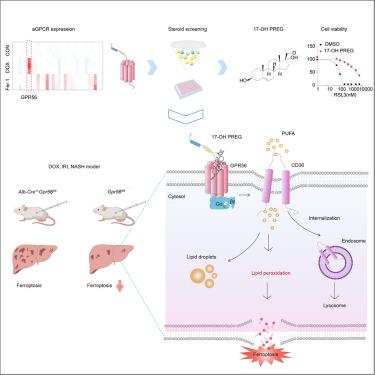
Sensing steroid hormone 17α-hydroxypregnenolone by GPR56 enables protection from ferroptosis-induced liver injury
G protein-coupled receptors (GPCRs) mediate most cellular responses to hormones, neurotransmitters, and environmental stimulants. However, whether GPCRs participate in tissue homeostasis through ferroptosis remains unclear. Here we identify that GPR56/ADGRG1 renders cells resistant to ferroptosis and deficiency of GPR56 exacerbates ferroptosis-mediated liver injury induced by doxorubicin (DOX) or ischemia-reperfusion (IR). Mechanistically, GPR56 decreases the abundance of phospholipids containing free polyunsaturated fatty acids (PUFAs) by promoting endocytosis-lysosomal degradation of CD36. By screening a panel of steroid hormones, we identified that 17α-hydroxypregnenolone (17-OH PREG) acts as an agonist of GPR56 to antagonize ferroptosis and efficiently attenuates liver injury before or after insult. Moreover, disease-associated GPR56 mutants were unresponsive to 17-OH PREG activation and insufficient to defend against ferroptosis. Together, our findings uncover that 17-OH PREG-GPR56 axis-mediated signal transduction works as a new anti-ferroptotic pathway to maintain liver homeostasis, providing novel insights into the potential therapy for liver injury.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Cell metabolism
生物-内分泌学与代谢
CiteScore
48.60
自引率
1.40%
发文量
173
审稿时长
2.5 months
期刊介绍:
Cell Metabolism is a top research journal established in 2005 that focuses on publishing original and impactful papers in the field of metabolic research.It covers a wide range of topics including diabetes, obesity, cardiovascular biology, aging and stress responses, circadian biology, and many others.
Cell Metabolism aims to contribute to the advancement of metabolic research by providing a platform for the publication and dissemination of high-quality research and thought-provoking articles.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


