凝集素结构域对人类 GnT-IV 家族活性的调控。
IF 2.4
3区 化学
Q3 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
N-Glycan 分支对糖蛋白的功能起着至关重要的调节作用,并与多种疾病有关。在参与 N-聚糖分支的糖基转移酶中,人类 N-乙酰葡糖胺基转移酶-IV(GnT-IV)家族有四个成员:该家族有四个成员:GnT-IVa、GnT-IVb、GnT-IVc 和 GnT-IVd。GnT-IVa 和 GnT-IVb 具有糖基转移酶活性,能在 N-聚糖的α1,3-曼臂上生成与 2 型糖尿病相关的 β1,4-GlcNAc分支,而 GnT-IVc 和 GnT-IVd 则没有这种活性。最近,人们发现该酶家族在 C 端区域有一个独特的凝集素结构域,该结构域对于酶在糖蛋白底物上的活性至关重要,但对于游离的 N-聚糖则不然。此外,GnT-IV 的凝集素结构域与附着在 GnT-IV 上的 N-糖之间的相互作用可实现 GnT-IV 活性的自我调节,这表明凝集素结构域在调控 GnT-IV 活性方面发挥着独特而关键的作用。在这篇综述中,我们总结了 GnT-IV 家族的生物学功能、对糖蛋白底物的选择性以及对酶活性的调控,重点是其独特的 C 端凝集素结构域。本文章由计算机程序翻译,如有差异,请以英文原文为准。
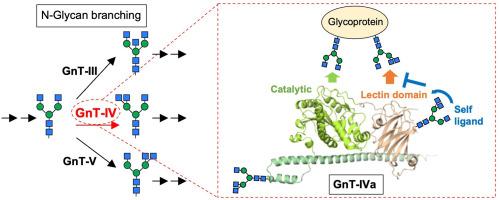
Regulation of human GnT-IV family activity by the lectin domain
N-Glycan branching critically regulates glycoprotein functions and is involved in various diseases. Among the glycosyltransferases involved in N-glycan branching is the human N-acetylglucosaminyltransferase-IV (GnT-IV) family, which has four members: GnT-IVa, GnT-IVb, GnT-IVc, and GnT-IVd. GnT-IVa and GnT-IVb have glycosyltransferase activity that generates the type-2 diabetes-related β1,4-GlcNAc branch on the α1,3-Man arm of N-glycans, whereas GnT-IVc and GnT-IVd do not. Recently, this enzyme family was found to have a unique lectin domain in the C-terminal region, which is essential for enzyme activity toward glycoprotein substrates but not toward free N-glycans. Furthermore, interaction between the lectin domain of GnT-IV and N-glycan attached to GnT-IV enables self-regulation of GnT-IV activity, indicating that the lectin domain plays a unique and pivotal role in the regulation of GnT-IV activity. In this review, we summarize the GnT-IV family's biological functions, selectivity for glycoprotein substrates, and regulation of enzymatic activity, with a focus on its unique C-terminal lectin domain.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Carbohydrate Research
化学-生化与分子生物学
CiteScore
5.00
自引率
3.20%
发文量
183
审稿时长
3.6 weeks
期刊介绍:
Carbohydrate Research publishes reports of original research in the following areas of carbohydrate science: action of enzymes, analytical chemistry, biochemistry (biosynthesis, degradation, structural and functional biochemistry, conformation, molecular recognition, enzyme mechanisms, carbohydrate-processing enzymes, including glycosidases and glycosyltransferases), chemical synthesis, isolation of natural products, physicochemical studies, reactions and their mechanisms, the study of structures and stereochemistry, and technological aspects.
Papers on polysaccharides should have a "molecular" component; that is a paper on new or modified polysaccharides should include structural information and characterization in addition to the usual studies of rheological properties and the like. A paper on a new, naturally occurring polysaccharide should include structural information, defining monosaccharide components and linkage sequence.
Papers devoted wholly or partly to X-ray crystallographic studies, or to computational aspects (molecular mechanics or molecular orbital calculations, simulations via molecular dynamics), will be considered if they meet certain criteria. For computational papers the requirements are that the methods used be specified in sufficient detail to permit replication of the results, and that the conclusions be shown to have relevance to experimental observations - the authors'' own data or data from the literature. Specific directions for the presentation of X-ray data are given below under Results and "discussion".
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


