主要拱顶蛋白整合了粘附驱动的信号,以调节胶原蛋白的重塑。
IF 4.4
2区 生物学
Q2 CELL BIOLOGY
引用次数: 0
摘要
DDR1 与纤维胶原相互作用,可影响β1整合素依赖性信号传导,但这两种不同受体之间功能性相互作用的介导机制尚未明确。我们寻找了连接 DDR1 和 b1 整合素依赖性信号转导与胶原蛋白结合的分子。与纤维胶原结合激活 DDR1 后,b1 整合素依赖的 ERK 磷酸化减少了 5 倍,而 ERK 磷酸化会导致 MMP1 的表达。相反,药物抑制 DDR1 或在纤维粘连蛋白上培养细胞可恢复 b1 整合素介导的 ERK 磷酸化和 MMP1 表达。磷酸化位点筛选表明,胶原诱导的 DDR1 激活通过调节焦点粘附激酶(FAK)的自身磷酸化,抑制了 β1 整合素依赖的 ERK 信号转导。免疫沉淀、质谱分析和蛋白质相互作用图谱显示,虽然DDR1和FAK不直接相互作用,但主要拱顶蛋白(MVP)会根据底物的不同与DDR1和FAK结合。在胶原蛋白上培养的表达 b1 整合素的细胞中,MVP 与 DDR1 相关。在胶原上培养的表达 DDR1 的细胞中,敲除 MVP 可恢复 ERK 的激活和 MMP1 的表达。人结肠浸润性癌症的免疫染色显示 DDR1 与 MVP 共定位。这些数据表明,MVP 与 DDR1 和 FAK 的相互作用有助于调节驱动胶原降解的 β1 整合素依赖性信号通路。本文章由计算机程序翻译,如有差异,请以英文原文为准。
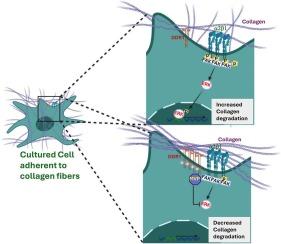
The major vault protein integrates adhesion-driven signals to regulate collagen remodeling
DDR1 interacts with fibrillar collagen and can affect β1 integrin-dependent signaling, but the mechanism that mediates functional interactions between these two different receptors is not defined. We searched for molecules that link DDR1 and β1 integrin-dependent signaling in response to collagen binding. The activation of DDR1 by binding to fibrillar collagen reduced by 5-fold, β1 integrin-dependent ERK phosphorylation that leads to MMP1 expression. In contrast, pharmacological inhibition of DDR1 or culturing cells on fibronectin restored ERK phosphorylation and MMP1 expression mediated by the β1 integrin. A phospho-site screen indicated that collagen-induced DDR1 activation inhibited β1 integrin-dependent ERK signaling by regulating autophosphorylation of focal adhesion kinase (FAK). Immunoprecipitation, mass spectrometry, and protein-protein interaction mapping showed that while DDR1 and FAK do not interact directly, the major vault protein (MVP) binds DDR1 and FAK depending on the substrate. MVP associated with DDR1 in cells expressing β1 integrin that were cultured on collagen. Knockdown of MVP restored ERK activation and MMP1 expression in DDR1-expressing cells cultured on collagen. Immunostaining of invasive cancers in human colon showed colocalization of DDR1 with MVP. These data indicate that MVP interactions with DDR1 and FAK contribute to the regulation of β1 integrin-dependent signaling pathways that drive collagen degradation.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Cellular signalling
生物-细胞生物学
CiteScore
8.40
自引率
0.00%
发文量
250
审稿时长
27 days
期刊介绍:
Cellular Signalling publishes original research describing fundamental and clinical findings on the mechanisms, actions and structural components of cellular signalling systems in vitro and in vivo.
Cellular Signalling aims at full length research papers defining signalling systems ranging from microorganisms to cells, tissues and higher organisms.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


