五环 N-杂环羰基二噻吩的高效合成及其光致变色特性
IF 1.5
4区 化学
Q3 CHEMISTRY, ORGANIC
引用次数: 0
摘要
鉴于光致变色功能材料具有出色的各向同性变色特性,这一领域正受到越来越多的关注。利用二噻吩位点实现光致变色的 N-杂环羰基光开关材料的开发通常涉及复杂的多步合成过程,其基底的多样化和制备过程的复杂性面临着各种挑战。在此,我们提出了一种简单的合成方法,通过使用 1,3-二苯基脲重新编程含二噻吩的咪唑啉酮的合成途径,制备光致变色的 4,5-双(2,5-二甲基噻吩-3-基)-1,3-二苯基-1,3-二氢-2H-咪唑-2-酮(1o)。目前的方法证明了α-羟基酮和 1,3-二苯基脲之间的高产率高效反应,从而实现了直接和简单的一步环化。目标 1o 的生长晶体样品经过了单晶 X 射线衍射分析,以阐明其结构信息。通过紫外线/可见光的交替照射,开环(o-异构体)和闭环(c-异构体)可毫不费力地相互转化,显示出显著的抗疲劳性和高对比度的视觉颜色变化。为了说明异构体的电子结构,我们基于 TD-DFT 计算对前沿分子轨道进行了深入分析。本文章由计算机程序翻译,如有差异,请以英文原文为准。
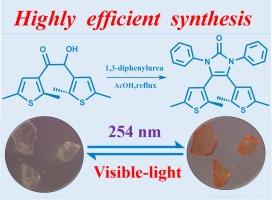
Highly efficient synthesis and photochromic properties of a pentacyclic N-heterocyclic carbonyl-based dithienylethene
Given that the photochromic functional materials possess outstanding phototropic color-change properties, the attention being paid to this field is progressively intensifying. The development of N-heterocyclic carbonyl photoswitching materials, harnessing dithienylethene site for photochromism, typically involves a complex multi-step synthesis process, which substrate diversification and the complexity of the preparation process are confronted with various challenges. Herein, we present a straightforward synthetic methodology for the preparation of photochromic 4,5-bis(2,5-dimethylthiophen-3-yl)-1,3-diphenyl-1,3-dihydro-2H-imidazol-2-one (1o) via reprogramming the synthesis pathway of dithienylethene-containing imidazolone using 1,3-diphenylurea. The current method demonstrates efficient reaction between α-hydroxy ketone and 1,3-diphenylurea in high yield, thereby enabling direct and straightforward one-step cyclization. The grown crystal sample of the targeted 1o has undergone single crystal X-ray diffraction analysis to elucidate its structural information. The ring-open (o-isomer) and ring-closed (c-isomer) forms showing remarkable fatigue resistance and high contrast visual color changes, can be effortlessly interconverted through the alternating irradiation of UV/vis light. To illustrate the electronic structure of the isomers, thorough analyses of the frontier molecular orbital based on TD-DFT calculations were conducted.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Tetrahedron Letters
化学-有机化学
CiteScore
3.50
自引率
5.60%
发文量
521
审稿时长
28 days
期刊介绍:
Tetrahedron Letters provides maximum dissemination of outstanding developments in organic chemistry. The journal is published weekly and covers developments in techniques, structures, methods and conclusions in experimental and theoretical organic chemistry. Rapid publication of timely and significant research results enables researchers from all over the world to transmit quickly their new contributions to large, international audiences.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


