富锂离子和锰离子纳米结构层状氧化物的机械电化学行为与硫化物全固态电池的优异容量保持率和电压衰减特性
IF 24.4
1区 材料科学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
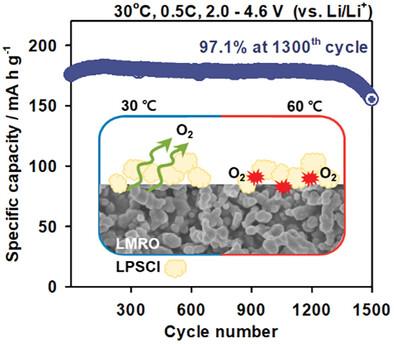
富含锂和锰的层状氧化物(LMROs)因其高比容量和高性价比而被公认为是锂离子电池(LIBs)的理想正极材料。然而,LMROs 也遇到了一些挑战,如电解质中的锰溶解和不可逆氧氧化还原反应释放出的氧气,导致结构退化和电压衰减,从而降低了能量密度。因此,最近的研究转向在全固态电池(ASSB)中使用 LMRO,因为在这种电池中锰的溶解可以忽略不计。在此,纳米结构 LMRO 与硫化物固体电解质在 ASSB 中的电化学相容性优于传统 LIB。纳米结构的 LMRO 具有出色的容量保持率(在 30 °C 下循环 1300 次后达到 97.1%),电压衰减显著降低。此外,还从复合阴极中机械-电化学相互作用的角度探讨了 LMRO 中 Li2MnO3 域的初始电化学活化。在高温条件下,由于 LMRO 释放出的氧气推动了 Li6PS5Cl 固体电解质的化学氧化,加速了界面降解。为解决这一问题,LMRO 表面通过酯化作用被硫代乙醇酸修饰,从而抑制了 Li6PS5Cl 的界面降解,并确保在 60 °C 下循环 500 次以上仍能保持稳定的容量。这些发现强调了 LMRO 材料作为 ASSB 负极选择的潜力,超过了用于 LIB 的材料。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Mechano-Electrochemical Behavior of Nanostructured Li- and Mn-Rich Layered Oxides with Superior Capacity Retention and Voltage Decay for Sulfide-Based All-Solid-State Batteries
Li- and Mn-rich layered oxides (LMROs) are recognized as promising cathode materials for lithium-ion batteries (LIBs) due to their high specific capacity and cost efficiency. However, LMROs encounter challenges such as manganese dissolution in electrolytes and the release of oxygen gas from irreversible oxygen redox reactions, leading to structural degradation and voltage decay that reduce energy density. Consequently, recent research has shifted toward employing LMROs in all-solid-state batteries (ASSBs), where Mn dissolution is negligible. Herein, nanostructured LMROs demonstrate superior electrochemical compatibility with sulfide-based solid electrolytes in ASSBs compared to conventional LIBs. Nanostructured LMRO exhibits outstanding capacity retention (97.1% after 1300 cycles at 30 °C) with significantly suppressed voltage decay. Furthermore, the initial electrochemical activation of Li2MnO3 domains within LMRO is explored in terms of the mechano-electrochemical interactions in the composite cathode. At elevated temperatures, interfacial degradation accelerates due to the chemical oxidation of Li6PS5Cl solid electrolytes, driven by oxygen released from LMRO. To address this, LMRO surfaces are modified with thioglycolic acid through esterification, suppressing interfacial degradation of Li6PS5Cl and ensuring stable capacity retention over 500 cycles at 60 °C. These findings underscore the potential of LMRO materials as promising cathode options for ASSBs, surpassing those used in LIBs.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Advanced Energy Materials
CHEMISTRY, PHYSICAL-ENERGY & FUELS
CiteScore
41.90
自引率
4.00%
发文量
889
审稿时长
1.4 months
期刊介绍:
Established in 2011, Advanced Energy Materials is an international, interdisciplinary, English-language journal that focuses on materials used in energy harvesting, conversion, and storage. It is regarded as a top-quality journal alongside Advanced Materials, Advanced Functional Materials, and Small.
With a 2022 Impact Factor of 27.8, Advanced Energy Materials is considered a prime source for the best energy-related research. The journal covers a wide range of topics in energy-related research, including organic and inorganic photovoltaics, batteries and supercapacitors, fuel cells, hydrogen generation and storage, thermoelectrics, water splitting and photocatalysis, solar fuels and thermosolar power, magnetocalorics, and piezoelectronics.
The readership of Advanced Energy Materials includes materials scientists, chemists, physicists, and engineers in both academia and industry. The journal is indexed in various databases and collections, such as Advanced Technologies & Aerospace Database, FIZ Karlsruhe, INSPEC (IET), Science Citation Index Expanded, Technology Collection, and Web of Science, among others.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


