Cu-Ce-a 纳米酶工程:模仿细胞色素 c 氧化酶的革命性合金纳米材料,用于超灵敏细胞色素 c 检测
IF 5.6
1区 化学
Q1 CHEMISTRY, ANALYTICAL
引用次数: 0
摘要
由于细胞色素 c 氧化酶(CcO)的结构复杂,包含多个金属修复位点和蛋白质亚基,因此设计细胞色素 c 氧化酶(CcO)的合成类似物是一项艰巨的任务。近年来,基于合金纳米材料的人工酶受到广泛关注,因为合金设计方法有望有效调整金属催化剂的特性。在本研究中,我们提出了铜铈合金纳米酶(Cu-Ce-a NEs),其中 Cu 模拟了 CcO 的活性位点,而 Ce 则赋予了合金相,并增强了催化氧化成细胞色素 c(Cyt c)的能力。Cu-Ce-a NEs 通过催化二氧到水的四电子还原,在功能上模拟了呼吸电子传递链(ETC)中的末端酶 CcO。利用 Cu-Ce-a NEs 的类 CcO 特性,我们成功地实现了对 Cyt c 的电化学检测。基于 Cu-Ce-a NEs 的电化学传感器显示出良好的线性范围,从 2 μM 到 20 μM Cyt c,检测限 (LOD) 为 2 μM。该方法对药物中 Cyt c 的定量具有很高的准确性,结果与实际浓度非常接近。这一发现不仅为酶类似物的设计提供了新的视角,而且强调了该方法在临床 Cyt c 检测方面的潜力,突出了它在生物医学研究和诊断方面的重要意义。本文章由计算机程序翻译,如有差异,请以英文原文为准。
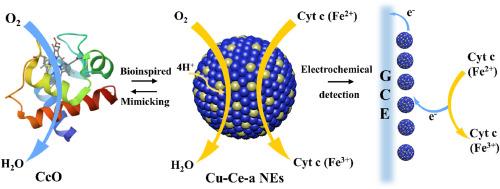
Engineering Cu–Ce-a nanozymes: Revolutionary alloy nanomaterials mimicking cytochrome c oxidase for ultra-sensitive cytochrome c detection
The design of synthetic analogs of cytochrome c oxidase (CcO) is a formidable task due to its intricate structure encompassing multiple metal prosthetic sites and protein subunits. In recent years, artificial enzymes based on alloy nanomaterials have garnered significant attention due to the alloy design approach holds promise for the effective tuning of the properties of metal catalysts. In this study, we present copper-cerium alloy nanozymes (Cu–Ce-a NEs), where Cu mimics the active site of CcO, while Ce endows the alloy phase and enhances the capacity to catalyze the oxidation to cytochrome c (Cyt c). Cu–Ce-a NEs functionally mimics CcO, the terminal enzyme in the respiratory electron transport chain (ETC), by catalyzing the four-electron reduction of dioxygen to water. Utilizing the CcO-like properties of Cu–Ce-a NEs, we successfully implemented the electrochemical detection of Cyt c. The Cu–Ce-a NEs based electrochemical sensor revealed a favorable linear range spanning from 2 to 20 μM Cyt c, with a detection limit (LOD) of 2 μM. This method demonstrates high accuracy in Cyt c quantitation in pharmaceuticals, with results closely aligning with the actual concentrations. This finding not only offers new perspectives in the design of enzyme analogs, but also underscores the potential of this method for clinical Cyt c detection, highlighting its significance in biomedical research and diagnostics.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Talanta
化学-分析化学
CiteScore
12.30
自引率
4.90%
发文量
861
审稿时长
29 days
期刊介绍:
Talanta provides a forum for the publication of original research papers, short communications, and critical reviews in all branches of pure and applied analytical chemistry. Papers are evaluated based on established guidelines, including the fundamental nature of the study, scientific novelty, substantial improvement or advantage over existing technology or methods, and demonstrated analytical applicability. Original research papers on fundamental studies, and on novel sensor and instrumentation developments, are encouraged. Novel or improved applications in areas such as clinical and biological chemistry, environmental analysis, geochemistry, materials science and engineering, and analytical platforms for omics development are welcome.
Analytical performance of methods should be determined, including interference and matrix effects, and methods should be validated by comparison with a standard method, or analysis of a certified reference material. Simple spiking recoveries may not be sufficient. The developed method should especially comprise information on selectivity, sensitivity, detection limits, accuracy, and reliability. However, applying official validation or robustness studies to a routine method or technique does not necessarily constitute novelty. Proper statistical treatment of the data should be provided. Relevant literature should be cited, including related publications by the authors, and authors should discuss how their proposed methodology compares with previously reported methods.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


