靶向 LINC00665/miR-199b-5p/SERPINE1 轴,通过 PI3K/AKt 通路抑制曲妥珠单抗抗性和胃癌的肿瘤发生
IF 5.9
3区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
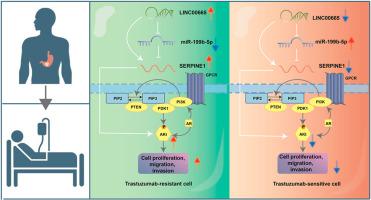
长非编码 RNA(lncRNA)是肿瘤进展和癌症耐药性的关键介质。在此,我们发现了一种与曲妥珠单抗耐药性和胃癌(GC)发展相关的lncRNA--LINC00665。LINC00665在胃癌组织中高表达,高表达与不良预后相关。经证实,敲除 LINC00665 可抑制胃癌的迁移、侵袭和对曲妥珠单抗的耐药性。此外,我们还发现LINC00665参与了幼稚B细胞、肥大细胞和T滤泡辅助细胞(Tfh)的浸润。从机理上讲,LINC00665被证实通过激活PI3K/AKt通路调控肿瘤发生和曲妥珠单抗抗性。LINC00665能使miR-199b-5p与SERPINE1的表达相互作用,导致AKt的磷酸化增加,从而参与PI3K/AKt通路。综上所述,LINC00665通过疏导miR-199b-5p和促进SERPINE1的表达,进一步激活了PI3K/AKt信号转导,从而促进了GC的肿瘤发生和曲妥珠单抗的耐药性;这一发现揭示了LINC00665调节GC肿瘤发生和耐药性的新机制。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Targeting LINC00665/miR-199b-5p/SERPINE1 axis to inhibit trastuzumab resistance and tumorigenesis of gastric cancer via PI3K/AKt pathway
Long noncoding RNAs (lncRNAs) serve as critical mediators of tumor progression and drug resistance in cancer. Herein, we identified a lncRNA, LINC00665, associated with trastuzumab resistance and development in gastric cancer (GC). LINC00665 was highly expressed in GC tissues and high expression of LINC00665 was correlated with poor prognosis. LINC00665 knockdown was verified to suppress migration, invasion, and resistance to trastuzumab in GC. Furthermore, we found that LINC00665 participates in the infiltration of naive B cells, mast cells, and T follicular helper (Tfh) cells. Mechanistically, LINC00665 was confirmed to regulate tumorigenesis and trastuzumab resistance by activating PI3K/AKt pathway. LINC00665 sponged miR-199b-5p to interact with SERPINE1 expression, resulting in the increase of phosphorylation of AKt, thus participating in the PI3K/AKt pathway. To summarize, LINC00665 facilitated the tumorigenesis and trastuzumab resistance of GC by sponging miR-199b-5p and promoting SERPINE1 expression, which further activated PI3K/AKt signaling; this finding reveals a new mechanism by which LINC00665 modulates tumor development and drug resistance in GC.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Non-coding RNA Research
Medicine-Biochemistry (medical)
CiteScore
7.70
自引率
6.00%
发文量
39
审稿时长
49 days
期刊介绍:
Non-coding RNA Research aims to publish high quality research and review articles on the mechanistic role of non-coding RNAs in all human diseases. This interdisciplinary journal will welcome research dealing with all aspects of non-coding RNAs-their biogenesis, regulation and role in disease progression. The focus of this journal will be to publish translational studies as well as well-designed basic studies with translational and clinical implications. The non-coding RNAs of particular interest will be microRNAs (miRNAs), small interfering RNAs (siRNAs), small nucleolar RNAs (snoRNAs), U-RNAs/small nuclear RNAs (snRNAs), exosomal/extracellular RNAs (exRNAs), Piwi-interacting RNAs (piRNAs) and long non-coding RNAs. Topics of interest will include, but not limited to: -Regulation of non-coding RNAs -Targets and regulatory functions of non-coding RNAs -Epigenetics and non-coding RNAs -Biological functions of non-coding RNAs -Non-coding RNAs as biomarkers -Non-coding RNA-based therapeutics -Prognostic value of non-coding RNAs -Pharmacological studies involving non-coding RNAs -Population based and epidemiological studies -Gene expression / proteomics / computational / pathway analysis-based studies on non-coding RNAs with functional validation -Novel strategies to manipulate non-coding RNAs expression and function -Clinical studies on evaluation of non-coding RNAs The journal will strive to disseminate cutting edge research, showcasing the ever-evolving importance of non-coding RNAs in modern day research and medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


