乙醇中碱介导的可见光驱动的喹喔啉-2(1H)-酮的 C-H 芳基化反应
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
之前利用可见光驱动 C-H 芳基化喹喔啉-2(1H)-酮的方法依赖于外部光催化剂。在此,我们报告了一种无光催化剂的芳基化方法。在这种方法中,β-二羰基碘鎓酰化物与乙醇中的 t-BuOK 结合,作为芳基前体,形成电子供体-受体 (EDA) 复合物。这些复合物能增强光吸收,促进有效的单电子转移和芳基自由基的形成。因此,各种喹喔啉-2(1H)-酮无需外加光催化剂即可进行精确高效的芳基化反应。该方案对各种官能团具有极佳的耐受性,反应条件温和,使用的溶剂环保,具有极高的生态价值。本文章由计算机程序翻译,如有差异,请以英文原文为准。
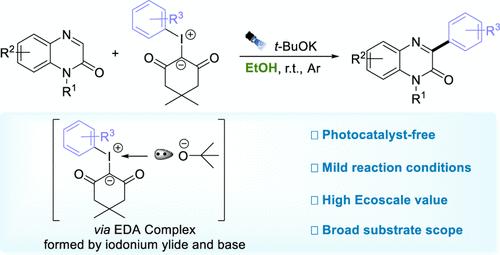
Base-Mediated Visible-Light-Driven C–H Arylation of Quinoxalin-2(1H)-Ones in Ethanol
Prior methods for visible-light-driven C–H arylation of quinoxalin-2(1H)-ones relied on external photocatalysts. Herein, we report a photocatalyst-free approach for this arylation. In this approach, β-dicarbonyl iodonium ylides, combined with t-BuOK in ethanol, act as aryl precursors, forming electron donor–acceptor (EDA) complexes. These complexes enhance light absorption, facilitating efficient single electron transfer and aryl radical formation. Consequently, various quinoxalin-2(1H)-ones undergo precise and efficient arylation without external photocatalysts. This protocol exhibits excellent tolerance toward diverse functional groups, with mild reaction conditions and eco-friendly solvents, revealing a high Ecoscale value.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


