首次展示体内 PDE11A4 靶点参与治疗老年性记忆障碍的潜力
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
PDE11A4 是治疗与年龄有关的记忆障碍的靶点。我们实验室之前的一份报告描述了一种代谢不稳定的强效、选择性 PDE11A4 抑制剂酰胺系列。对易变酰胺分子的杂环酰胺异构体的研究揭示了独特的结构-活性关系,并发现了几种效力与酰胺系列相当的化合物。本手稿描述了这组化合物的结构-活性和结构-性质关系特征,从而鉴定出了一种口服生物可用性、脑穿透性、选择性和强效 PDE11A4 抑制剂。靶点参与实验证明了 PDE11A4 对小鼠下丘脑的抑制作用,而 PDE11A4 基因敲除动物则没有这种抑制作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
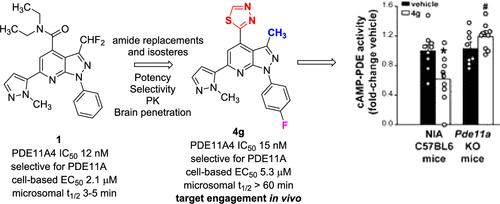
First Demonstration of In Vivo PDE11A4 Target Engagement for Potential Treatment of Age-Related Memory Disorders
PDE11A4 is a target of interest for the treatment of age-related memory disorders. A previous report from our laboratories described an amide series of potent, selective PDE11A4 inhibitors that was metabolically unstable. Investigation of heterocyclic amide isosteres for the labile amide moiety revealed distinct structure–activity relationships and identified several compounds with potency comparable to the amide series. This manuscript describes the characterization of structure–activity and structure–property relationships in this set, leading to the identification of an orally bioavailable, brain-penetrant, selective and potent PDE11A4 inhibitor. Target engagement experiments demonstrated PDE11A4 inhibition in the hypothalamus of mice that was absent in PDE11A4 knock out animals.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


