原子效率高的原药方法使 H2S 和非甾体抗炎药产生协同作用并提高安全性
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
由于硫化氢(H2S)的结构实体复杂且释放动力学独立,因此硫化氢与非甾体抗炎药协同作用的努力面临着挑战。本研究提出了一种原子效率极高的方法,即使用硫代羧酸(硫酸)作为新型 H2S 释放前体,并成功地将其用于修饰非甾体抗炎药,该方法具有几个关键优势。首先,硫代羧酸修饰的非甾体抗炎药具有抑制环氧化酶的活性,有时还能提高药效。其次,这种原药方法避免了引入额外的结构分子,只允许释放预期的活性成分。第三,H2S 和非甾体抗炎药的释放是同时进行的,从而使两种活性成分的浓度分布达到最佳同步。该设计基于我们的发现,即酯酶可以直接有效地水解硫代羧酸,从而实现 H2S 的控制释放。这项研究通过合成类似物证明了原理,评估了释放动力学、酶抑制和药效,并在动物模型中评估了毒性和肠道微生物群调节。本文章由计算机程序翻译,如有差异,请以英文原文为准。
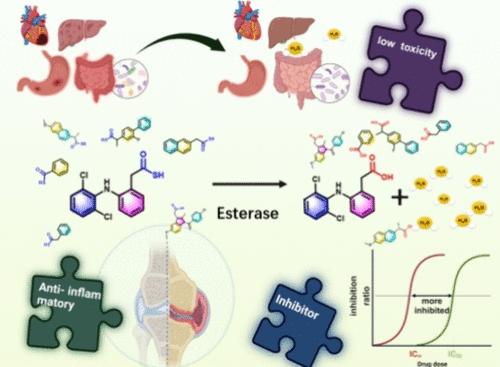
A Highly Atom-Efficient Prodrug Approach to Generate Synergy between H2S and Nonsteroidal Anti-inflammatory Drugs and Improve Safety
Efforts to synergize hydrogen sulfide (H2S) with NSAIDs have faced challenges due to complex structural entities and independent release kinetics. This study presents a highly atom-efficient approach of using a thiocarboxylic acid (thioacid) as a novel H2S releasing precursor and successfully employs it to modify NSAIDs, which offers several critical advantages. First, thioacid-modified NSAID is active in inhibiting cyclooxygenase, sometimes with improved potency. Second, this prodrug approach avoids introducing extra structural moieties, allowing for the release of only the intended active principals. Third, the release of H2S and NSAID is concomitant, thus optimally synchronizing the concentration profiles of the two active principals. The design is based on our discovery that esterases can directly and efficiently hydrolyze thiocarboxylic acids, enabling controlled release H2S. This study demonstrates the proof of principle through synthesizing analogs, assesses release kinetics, enzyme inhibition, and pharmacological efficacy, and evaluates toxicity and gut microbiota regulation in animal models.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


