用于靶向成纤维细胞活化蛋白的 68Ga/177Lu 标记放射治疗对,可提高肿瘤吸收和保留率
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
成纤维细胞活化蛋白(FAP)在90%以上的肿瘤的癌相关成纤维细胞上特异性表达,被认为是癌症治疗的一个有前途的靶点。在此,我们开发了一种新型示踪剂 DOTA-FAPT,并用镓-68 和镥-177 将其标记为一对治疗剂。[68Ga]Ga/[177Lu]Lu-FAPT具有高稳定性和亲水性,并与FAP靶点有很强的亲和力。显微PET/CT成像显示,与[68Ga]Ga-FAPI-04相比,[68Ga]Ga-FAPT在肿瘤中的摄取量明显增加,在A549-FAP和U87MG肿瘤异种移植物中的保留时间延长,显示出良好的体内药代动力学特性。治疗研究表明,与[177Lu]Lu-FAPI-04相比,[177Lu]Lu-FAPT具有更高的肿瘤蓄积性,从而产生更强的肿瘤生长抑制作用。首次人体评估还显示,[68Ga]Ga-FAPT 在一名肺癌患者体内分布良好,对原发灶和淋巴结转移灶具有卓越的诊断效果。令人鼓舞的结果表明,68Ga/177Lu 标记的 DOTA-FAPT 是一种具有广阔应用前景的治疗药物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
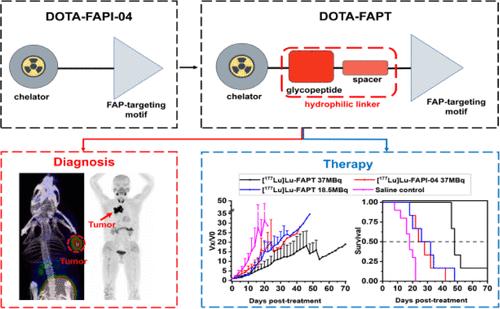
68Ga/177Lu-Labeled Theranostic Pair for Targeting Fibroblast Activation Protein with Improved Tumor Uptake and Retention
Fibroblast activation protein (FAP) is specifically expressed on cancer-associated fibroblasts in over 90% of tumors and is considered a promising target for cancer theranostics. Here, we developed a novel tracer, DOTA-FAPT, and labeled it with gallium-68 and lutetium-177 as a theranostic pair. [68Ga]Ga/[177Lu]Lu-FAPT exhibited high stability and hydrophilicity, as well as strong affinity to the FAP target. Micro-PET/CT imaging revealed that [68Ga]Ga-FAPT exhibited significantly increased uptake in tumors and extended retention in A549-FAP and U87MG tumor xenografts as compared to [68Ga]Ga-FAPI-04, demonstrating favorable pharmacokinetic characteristics in vivo. Therapeutic studies showed that [177Lu]Lu-FAPT had higher tumor accumulation compared to [177Lu]Lu-FAPI-04, leading to stronger tumor growth inhibition. The first-in-human evaluation also revealed that [68Ga]Ga-FAPT has good in vivo distribution and superior diagnostic efficacy on primary and lymph node metastases in a patient with lung cancer. Our encouraging results suggest that 68Ga/177Lu-labeled DOTA-FAPT is a theranostic pair with broad application prospect.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


