在工业级碱性条件下,卟啉钙化 NiFe-LDH 对铁进行配位稳定,以实现长期电催化水氧化
IF 18.5
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
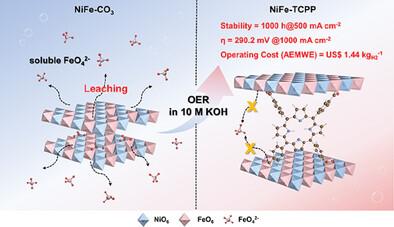
在工业碱性条件下具有高电流密度的耐用且经济的电催化剂,对于推进水电解氢能的工业生产至关重要。工业高碱性电解质会加剧镍铁层双氢氧化物(NiFe-LDH)的铁溶解,导致稳定性和活性急剧下降。本研究分别制备了夹杂四(4-羧基苯基)卟啉(TCPP)(NiFe-TCPP)、1,4,7,10-四氮杂环十二烷-1,4,7,10-四乙酸(DOTA)(NiFe-DOTA)和 CO32-(NiFe-CO3)的镍铁层状双氢氧化物,用于工业碱性条件下的电催化水氧化。在 10 m KOH 中,与 NiFe-DOTA(335.0 mV)和 NiFe-CO3(499.2 mV)相比,制得的 NiFe-TCPP 在 1000 mA cm-2 时的过电位最低,为 290.2 mV。NiFe-TCPP 还能在 500 mA cm-2 下连续工作 1000 小时,衰减几乎为零。理论和实验研究表明,共轭羧酸配体 TCPP 与层压板中的铁之间的强配位通过将溶解能垒提高到 4.29 eV 来抑制铁的浸出,并提高了自修复能力,从而增强了稳定性。此外,强配位引起的电荷再分布优化了 d 带中心(-2.81 eV),降低了反应能垒(1.47 eV),从而提高了催化活性。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Coordination Stabilization of Fe by Porphyrin-Intercalated NiFe-LDH Under Industrial-Level Alkaline Conditions for Long-Term Electrocatalytic Water Oxidation
The durable and economic electrocatalysts with high current density under industrial alkaline conditions are critical for advancing the industrial production of hydrogen energy by water electrolysis. The industrial highly alkaline electrolyte exacerbates the Fe dissolution of NiFe layered double hydroxide (NiFe-LDH), leading to the dramatic degradation of stability and activity. The NiFe-LDH intercalated Tetrakis(4-carboxyphenyl)porphyrin (TCPP) (NiFe-TCPP), 1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) (NiFe-DOTA) and CO32− (NiFe-CO3) are fabricated respectively for electrocatalytic water oxidation under industrial alkaline conditions. In 10 m KOH, compared to NiFe-DOTA (335.0 mV) and NiFe-CO3 (499.2 mV), the resultant NiFe-TCPP exhibits the lowest overpotentials of 290.2 mV at 1000 mA cm−2. The NiFe-TCPP also operates continuously for 1000 h at 500 mA cm−2 with near-zero attenuation. The theoretical and experimental studies reveal that the strong coordination between conjugated carboxylate ligand TCPP and Fe of the laminate inhibits the Fe leaching by increasing the dissolution energy barriers to 4.29 eV and improving the self-healing ability, thus enhancing the stability. Furthermore, the charge redistribution induced by the strong coordination optimizes the d-band centers (-2.81 eV) and decreases the reaction energy barriers (1.47 eV), thereby increasing the catalytic activity.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Advanced Functional Materials
工程技术-材料科学:综合
CiteScore
29.50
自引率
4.20%
发文量
2086
审稿时长
2.1 months
期刊介绍:
Firmly established as a top-tier materials science journal, Advanced Functional Materials reports breakthrough research in all aspects of materials science, including nanotechnology, chemistry, physics, and biology every week.
Advanced Functional Materials is known for its rapid and fair peer review, quality content, and high impact, making it the first choice of the international materials science community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


