合成修饰寡核苷酸过程中来自羟脯氨酸衍生物的杂质
IF 3.1
3区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
摘要
本研究探讨了合成修饰寡核苷酸所面临的挑战,尤其关注使用羟脯氨酸衍生物时引入的杂质。我们发现,加入氨基连接体,特别是反式-4-羟基-l-脯氨醇骨架,会导致形成难以去除的特定杂质。为了解决这些问题,我们建议在固相载体上预装连接体,以防止产生磷酰胺杂质。此外,我们还建议在裂解和脱保护步骤中使用清除剂,以尽量减少不必要的反应。一个重要发现是三乙胺在洗涤步骤中的关键作用,增加三乙胺的用量可有效防止丙烯腈和乙酰化加合物的形成。通过一系列实验,我们找出了产生这些杂质的原因,并提出了减少杂质的策略。这些策略可确保高效合成纯度更高的寡核苷酸,提高其在生物医学研究和生物技术领域的广泛应用潜力。本文章由计算机程序翻译,如有差异,请以英文原文为准。
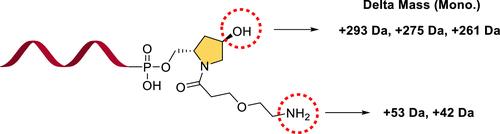
Impurities from Hydroxyproline Derivatives in the Synthesis of Modified Oligonucleotides
This study explores the challenges of synthesizing modified oligonucleotides, particularly focusing on the impurities introduced when using hydroxyproline derivatives. We found that incorporating amino linkers, specifically the trans-4-hydroxy-l-prolinol backbone, leads to the formation of specific difficult-to-remove impurities. To address these issues, we recommend preloading the linker onto the solid-phase carrier to prevent the generation of phosphoramide impurities. Additionally, using a scavenger during the cleavage and deprotection steps is suggested to minimize unwanted reactions. A significant finding is the crucial role of triethylamine in the washing steps, where increasing its amount effectively prevents the formation of acrylonitrile and acetylation adducts. Through a series of experiments, we have identified the causes of these impurities and proposed strategies for their mitigation. These strategies ensure the efficient synthesis of oligonucleotides with improved purity, enhancing their potential for broader applications in biomedical research and biotechnology.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
6.90
自引率
14.70%
发文量
251
审稿时长
2 months
期刊介绍:
The journal Organic Process Research & Development serves as a communication tool between industrial chemists and chemists working in universities and research institutes. As such, it reports original work from the broad field of industrial process chemistry but also presents academic results that are relevant, or potentially relevant, to industrial applications. Process chemistry is the science that enables the safe, environmentally benign and ultimately economical manufacturing of organic compounds that are required in larger amounts to help address the needs of society. Consequently, the Journal encompasses every aspect of organic chemistry, including all aspects of catalysis, synthetic methodology development and synthetic strategy exploration, but also includes aspects from analytical and solid-state chemistry and chemical engineering, such as work-up tools,process safety, or flow-chemistry. The goal of development and optimization of chemical reactions and processes is their transfer to a larger scale; original work describing such studies and the actual implementation on scale is highly relevant to the journal. However, studies on new developments from either industry, research institutes or academia that have not yet been demonstrated on scale, but where an industrial utility can be expected and where the study has addressed important prerequisites for a scale-up and has given confidence into the reliability and practicality of the chemistry, also serve the mission of OPR&D as a communication tool between the different contributors to the field.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


