疏水性固体脂质微粒,用于保护胃敏感的亲水性活性生物分子,口服治疗 EPI。
IF 4.4
2区 医学
Q1 PHARMACOLOGY & PHARMACY
European Journal of Pharmaceutics and Biopharmaceutics
Pub Date : 2024-09-18
DOI:10.1016/j.ejpb.2024.114504
引用次数: 0
摘要
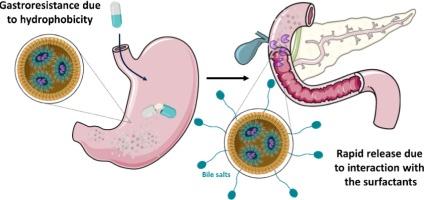
由囊性纤维化、慢性胰腺炎和克罗恩病等疾病引起的胰腺外分泌功能不全(EPI)是一种经常被忽视和诊断不足的胃肠道疾病。它因胰液分泌不足而导致肠道消化功能不全,引起不适、疼痛,最终导致严重营养不良。尽管在过去 30 年中有许多治疗方法被证明无效,但本研究提出了一种严格的疏水性固体脂质配方,通过口服给药来恢复消化功能。该技术依靠基质的疏水性来保护亲水性活性成分免受胃环境的影响,同时通过针对胆汁盐的两亲性使其在十二指肠中立即释放。研究结果表明,这种制剂能在胃液(模拟胃液或 SGF)中有效保护对酸敏感的活性成分,使其在进入人工十二指肠环境(模拟肠液或 SIF)后迅速释放。此外,有研究表明,蛋白质类活性成分的保存不仅限于其主要蛋白质结构,还包括其功能方面,如酶活性。这种给药技术可以保护胰蛋白酶等对胃酸敏感的亲水性活性生物大分子,同时促进其在十二指肠近端与胆盐接触后立即释放,最终达到纠正 EPI 引起的消化缺陷的目的。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Hydrophobic solid lipid-based microparticles for the protection of gastric-sensitive hydrophilic active biomolecules for oral administration in the treatment of EPI
Exocrine Pancreatic Insufficiency (EPI), induced by conditions such as cystic fibrosis, chronic pancreatitis, and Crohn’s disease, is a frequently overlooked and underdiagnosed gastrointestinal disorder. It leads to inadequate intestinal digestion due to insufficient secretion of pancreatic juice, resulting in discomfort, pain, and ultimately severe malnutrition. Despite numerous treatments proving ineffective over the past three decades, a strictly hydrophobic solid lipid formulation, administered orally, is proposed in this study to restore digestive function. This technology relies on the hydrophobic nature of the matrix to physically protect the hydrophilic active principle from the gastric environment while enabling its immediate release in the duodenum by targeting the amphiphilic nature of bile salts. Results demonstrate that this formulation effectively protects an acid-sensitive active ingredient during gastric passage (Simulated Gastric Fluid or SGF), facilitating its rapid release upon entering an artificial duodenal environment (Simulated Intestinal Fluid or SIF). Furthermore, it has been demonstrated that the preservation of a protein-based active ingredient extends beyond its primary protein structure to include its functional aspects, such as enzymatic activity. This drug delivery technology could enable the protection of hydrophilic active biomolecules, such as pancreatin, which are sensitive to gastric acidity, while promoting their immediate release upon contact with bile salts in the proximal duodenum, with the ultimate goal of correcting the digestive defect induced by EPI.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
8.80
自引率
4.10%
发文量
211
审稿时长
36 days
期刊介绍:
The European Journal of Pharmaceutics and Biopharmaceutics provides a medium for the publication of novel, innovative and hypothesis-driven research from the areas of Pharmaceutics and Biopharmaceutics.
Topics covered include for example:
Design and development of drug delivery systems for pharmaceuticals and biopharmaceuticals (small molecules, proteins, nucleic acids)
Aspects of manufacturing process design
Biomedical aspects of drug product design
Strategies and formulations for controlled drug transport across biological barriers
Physicochemical aspects of drug product development
Novel excipients for drug product design
Drug delivery and controlled release systems for systemic and local applications
Nanomaterials for therapeutic and diagnostic purposes
Advanced therapy medicinal products
Medical devices supporting a distinct pharmacological effect.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


