镍烯丙基络合物中的叔丁基和三甲基硅基取代基:相似但不相同
IF 3.3
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
具有 t-Bu 取代烯丙基配体的金属配合物相对罕见,尤其是与概念上相似的三甲基硅取代类似物相比。这种稀缺性部分源于 t-Bu 版本的一般合成切入点很少。我们通过对烯丙基配体本身进行改良合成,并形成几种单(烯丙基)镍衍生物,对这种情况进行了研究。将 2,2,6,6- 四甲基-4-庚烯-3-酮转化为相关的 5-溴-2,2,6,6-四甲基庚-3-烯(A2tBr)后,发现在中性供体配体(如 MeCN)存在下,Ni(COD)2 和 A2tBr 的混合物会生成暗红色的二聚 π-烯丙基络合物 [{A2tNiBr}2]。核磁共振和 X 射线晶体学数据都证实,t-Bu 取代基与之前描述的[{A′NiBr}2](A′=1,3-(TMS)2C3H3)复合物中的 t-Bu 取代基一样,呈 syn、syn-构象。{A2tNiBr}2]会与 PPh3 和 IMes(IMes = 1,3-二甲基咪唑-2-亚基)等中性供体形成加合物,但生成的[A2tNi(PPh3)Br]复合物不如其三甲基硅类似物稳定。[A2tNi(IMes)Br]复合物以单体形式从己烷中结晶出来,具有η3配位的[A2t]配体,与[{A2tNiBr}2]中的起始排列不同,A2t配体上的t-Bu基团呈同位反向关系。这种结构与三甲基硅类似物[A′Ni(IMes)Br]类似。DFT 计算用于比较 t-Bu 基团和相关的三甲基硅取代配合物的结构。本文章由计算机程序翻译,如有差异,请以英文原文为准。
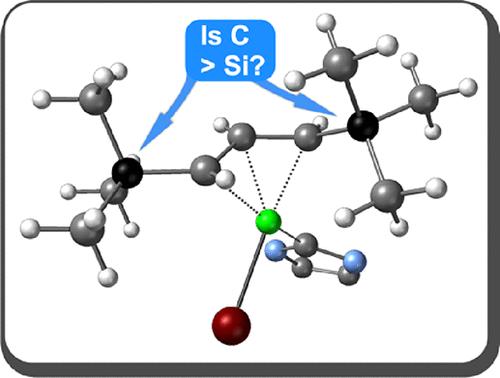
t-Butyl and Trimethylsilyl Substituents in Nickel Allyl Complexes: Similar but Not the Same
Metal complexes with t-Bu-substituted allyl ligands are relatively rare, especially compared to their conceptually similar trimethylsilyl-substituted analogs. The scarcity partially stems from the few general synthetic entry points for the t-Bu versions. This situation was studied through a modified synthesis for the allyl ligand itself and by forming several mono(allyl)nickel derivatives. After 2,2,6,6-tetramethyl-4-hepten-3-one was converted to the related 5-bromo-2,2,6,6-tetramethylhept-3-ene (A2tBr), a mixture of Ni(COD)2 and A2tBr in the presence of a neutral donor ligand such as MeCN was found to produce the dark red dimeric π-allyl complex [{A2tNiBr}2]. Both NMR and X-ray crystallographic data confirmed that the t-Bu substituents are in a syn, syn-conformation, like that in the previously described [{A′NiBr}2] (A′ = 1,3-(TMS)2C3H3) complex. [{A2tNiBr}2] will form adducts with neutral donors such as PPh3 and IMes (IMes = 1,3-dimesitylimidazol-2-ylidene), but the resulting [A2tNi(PPh3)Br] complex is not as stable as its trimethylsilyl analog. The [A2tNi(IMes)Br] complex crystallizes from hexanes as a monomer, with an η3-coordinated [A2t] ligand, and in contrast to the starting arrangement in [{A2tNiBr}2], the t-Bu groups on the A2t ligand are in a syn, anti-relationship. This structure is paralleled in the trimethylsilyl analog [A′Ni(IMes)Br]. DFT calculations were used to compare the structures of t-Bu- and related trimethylsilyl-substituted complexes.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Organic & Inorganic Au
有机化学、无机化学-
CiteScore
4.10
自引率
0.00%
发文量
0
期刊介绍:
ACS Organic & Inorganic Au is an open access journal that publishes original experimental and theoretical/computational studies on organic organometallic inorganic crystal growth and engineering and organic process chemistry. Short letters comprehensive articles reviews and perspectives are welcome on topics that include:Organic chemistry Organometallic chemistry Inorganic Chemistry and Organic Process Chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


