熔盐调制钾氮碳以打破光催化整体水分离的动力学瓶颈并减少对环境的影响
IF 15.8
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
缓慢的界面水解离和 O2 演化反应(OER)动力学是限制光催化整体水分离性能的主要障碍。本文展示了钾-氮-碳的熔盐调制,形成了高结晶的钾掺杂聚(三嗪亚胺)(KPTI)。现场 X 射线衍射图样和理论计算表明,由于形成了动力学稳定的 KPTI,氯化钾熔体可显著降低三嗪结构单元缩聚的自由能。得益于钾-碳-氮分子的存在,催化剂不仅能削弱激子束缚,提高光生电荷载流子的分离效率,还能通过抑制不希望的 C═O 的形成来增强碳位点的稳定性。此外,KPTI 还能通过形成具有有序结构的界面 K-H2O 簇来加速水的解离,从而为 H2 演化反应提供充足的质子,并降低能垒以增强 OER 的动力学。因此,在 KPTI 上可以实现稳定的光催化整体水分离性能,并按化学计量生成产物(H2 和 O2)。生命周期评估表明,随着电网中绿色电力的增加,KPTI 生产可在短期内实现碳中和。本文章由计算机程序翻译,如有差异,请以英文原文为准。
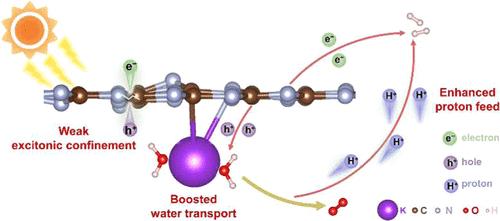
Molten Salt Modulation of Potassium–Nitrogen–Carbon for the Breaking Kinetics Bottleneck of Photocatalytic Overall Water Splitting and Environmental Impact Reduction
Sluggish interfacial water dissociation and the O2 evolution reaction (OER) kinetics are the main obstacles that limit the photocatalytic overall water-splitting performance. A molten salt modulation of potassium–nitrogen–carbon is herein demonstrated as the formation of highly crystalline potassium-doped poly(triazine imide) (KPTI). The in situ X-ray diffraction patterns and theoretical calculation show that the KCl melt can significantly reduce the free energy for the polycondensation of triazine building blocks owing to the formation of a kinetically stable KPTI. Benefiting from the presence of potassium–carbon–nitrogen moiety, the catalyst not only weakens the excitonic confinement to improve the separation efficiency of photogenerated charge carriers but also enhances the stability of carbon sites by suppressing the undesired C═O formation. Moreover, KPTI accelerates water dissociation by forming interfacial K·H2O clusters with an ordered structure, which supplies sufficient protons for the H2 evolution reaction and lowers the energy barrier to enhance the kinetics of OER. Therefore, a stable photocatalytic overall water-splitting performance can be achieved over KPTI with a stoichiometric generation of products (H2 and O2). Life cycle assessment shows that a carbon-neutral scenario can be achieved on KPTI production in the near term with an increase in green power in the electricity grid.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Nano
工程技术-材料科学:综合
CiteScore
26.00
自引率
4.10%
发文量
1627
审稿时长
1.7 months
期刊介绍:
ACS Nano, published monthly, serves as an international forum for comprehensive articles on nanoscience and nanotechnology research at the intersections of chemistry, biology, materials science, physics, and engineering. The journal fosters communication among scientists in these communities, facilitating collaboration, new research opportunities, and advancements through discoveries. ACS Nano covers synthesis, assembly, characterization, theory, and simulation of nanostructures, nanobiotechnology, nanofabrication, methods and tools for nanoscience and nanotechnology, and self- and directed-assembly. Alongside original research articles, it offers thorough reviews, perspectives on cutting-edge research, and discussions envisioning the future of nanoscience and nanotechnology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


