铜/钯催化的α-溴羰基串联羰基合成含酯的 3,4-二氢喹啉-2(1H)-酮衍生物
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
为了加速构建含酯的 3,4-二氢喹啉-2(1H)-酮支架,我们开发了一种铜/钯催化的串联自由基环化和烷基系α-溴羰基与酚的羰基化反应。通过使用苯-1,3,5-三甲酸苯酯(TFBen)作为 CO 源,反应顺利进行,并以高产率获得了一系列含酯的 3,4-二氢喹啉-2(1H)-酮衍生物。此外,这种方法还可用于生物活性分子的后期修饰。本文章由计算机程序翻译,如有差异,请以英文原文为准。
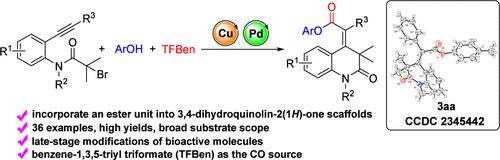
Cu/Pd-Catalyzed Tandem Carbonylative Synthesis of Ester-Containing 3,4-Dihydroquinolin-2(1H)-one Derivatives from Alkyne-Tethered α-Bromocarbonyls
A Cu/Pd-catalyzed tandem radical cyclization and carbonylation of alkyne-tethered α-bromocarbonyls with phenols has been developed for the expedited construction of ester-containing 3,4-dihydroquinolin-2(1H)-one scaffolds. By employing benzene-1,3,5-triyl triformate (TFBen) as the CO source, the reaction proceeded smoothly to afford a series of ester-containing 3,4-dihydroquinolin-2(1H)-one derivatives in high yields. Moreover, this method could be utilized in the late-stage modifications of bioactive molecules.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


