新辅助尼妥珠单抗或尼妥珠单抗加伊匹单抗治疗早期三阴性乳腺癌:2期适应性试验
IF 58.7
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
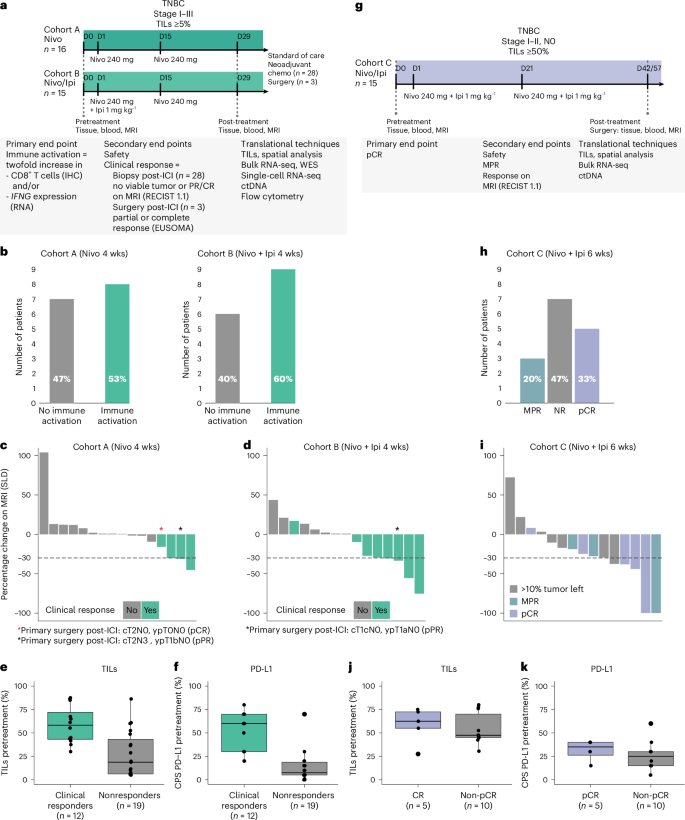
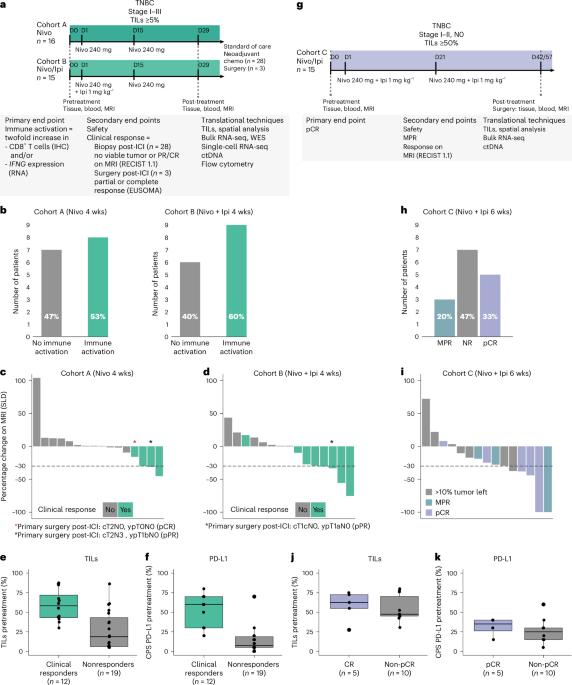
免疫检查点抑制剂(ICI)联合化疗是目前治疗II-III期三阴性乳腺癌的标准疗法;然而,对于哪些患者可以选择不化疗的ICI,以及联合ICI的益处有多大,目前还不得而知。适应性 BELLINI 试验探讨了短期联合 ICI 是否会诱导免疫激活(主要终点:CD8+ T 细胞或 IFNG 增加两倍),为不进行化疗的新辅助 ICI 提供了理论依据。在这里,我们在机会之窗队列A(4周抗PD-1)和队列B(4周抗PD-1+抗CTLA4)中分别观察到53%(15例中的8例)和60%(15例中的9例)的患者出现免疫激活。高水平的肿瘤浸润淋巴细胞与反应相关。单细胞 RNA 测序显示,治疗前较高的肿瘤反应性 CD8+ T 细胞、滤泡辅助性 T 细胞以及肿瘤与 CD8+ T 细胞之间较短的距离与反应相关。治疗后较高水平的调节性T细胞与无应答相关。基于这些数据,我们为肿瘤浸润淋巴细胞水平较高(≥50%)的患者设立了队列C,这些患者接受了6周的新辅助抗PD-1+抗CTLA4治疗,随后进行了手术(主要终点:病理完全反应)。总体而言,53%的患者(15例中的8例)在切除时获得了主要病理反应(肿瘤存活率为10%),33%的患者(15例中的5例)获得了病理完全反应。所有组别均符合西蒙的两期阈值,可扩展至 II 期。我们观察到17%的患者出现≥3级不良反应,免疫介导的内分泌病变发生率较高(57%)。总之,无化疗的新辅助免疫疗法具有潜在疗效,值得对早期三阴性乳腺癌患者进行进一步研究。ClinicalTrials.gov 注册:NCT03815890。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Neoadjuvant nivolumab or nivolumab plus ipilimumab in early-stage triple-negative breast cancer: a phase 2 adaptive trial
Immune checkpoint inhibition (ICI) with chemotherapy is now the standard of care for stage II–III triple-negative breast cancer; however, it is largely unknown for which patients ICI without chemotherapy could be an option and what the benefit of combination ICI could be. The adaptive BELLINI trial explored whether short combination ICI induces immune activation (primary end point, twofold increase in CD8+ T cells or IFNG), providing a rationale for neoadjuvant ICI without chemotherapy. Here, in window-of-opportunity cohorts A (4 weeks of anti-PD-1) and B (4 weeks of anti-PD-1 + anti-CTLA4), we observed immune activation in 53% (8 of 15) and 60% (9 of 15) of patients, respectively. High levels of tumor-infiltrating lymphocytes correlated with response. Single-cell RNA sequencing revealed that higher pretreatment tumor-reactive CD8+ T cells, follicular helper T cells and shorter distances between tumor and CD8+ T cells correlated with response. Higher levels of regulatory T cells after treatment were associated with nonresponse. Based on these data, we opened cohort C for patients with high levels of tumor-infiltrating lymphocytes (≥50%) who received 6 weeks of neoadjuvant anti-PD-1 + anti-CTLA4 followed by surgery (primary end point, pathological complete response). Overall, 53% (8 of 15) of patients had a major pathological response (<10% viable tumor) at resection, with 33% (5 of 15) having a pathological complete response. All cohorts met Simon’s two-stage threshold for expansion to stage II. We observed grade ≥3 adverse events for 17% of patients and a high rate (57%) of immune-mediated endocrinopathies. In conclusion, neoadjuvant immunotherapy without chemotherapy demonstrates potential efficacy and warrants further investigation in patients with early triple-negative breast cancer. ClinicalTrials.gov registration: NCT03815890 . In the phase 2 adaptive BELLINI trial, patients with early-stage triple-negative breast cancer received neoadjuvant nivolumab with or without ipilimumab, showing immune activation, clearance of circulating tumor DNA and promising clinical response rates, especially in patients preselected based on high levels of tumor-infiltrating lymphocytes.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Medicine
医学-生化与分子生物学
CiteScore
100.90
自引率
0.70%
发文量
525
审稿时长
1 months
期刊介绍:
Nature Medicine is a monthly journal publishing original peer-reviewed research in all areas of medicine. The publication focuses on originality, timeliness, interdisciplinary interest, and the impact on improving human health. In addition to research articles, Nature Medicine also publishes commissioned content such as News, Reviews, and Perspectives. This content aims to provide context for the latest advances in translational and clinical research, reaching a wide audience of M.D. and Ph.D. readers. All editorial decisions for the journal are made by a team of full-time professional editors.
Nature Medicine consider all types of clinical research, including:
-Case-reports and small case series
-Clinical trials, whether phase 1, 2, 3 or 4
-Observational studies
-Meta-analyses
-Biomarker studies
-Public and global health studies
Nature Medicine is also committed to facilitating communication between translational and clinical researchers. As such, we consider “hybrid” studies with preclinical and translational findings reported alongside data from clinical studies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


