趋化因子受体 CXCR4 与 CXCL12 和 hBD-3 的相互作用和动态结合
IF 6.2
2区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
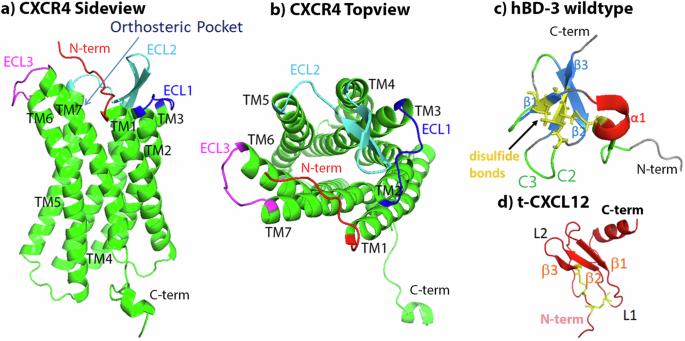
趋化因子受体 CXCR4 与多种疾病有关。基于总共 63 µs 的全原子 MD 模拟,我们对嵌入 POPC 脂质双分子层的 CXCR4 与完整和截断形式的 CXCL12 以及野生型、类似物和突变型 hBD-3 的结合进行了比较研究。利用 HADDOCK docking 或随机种子方法预测了 CXCR4 与配体的初始结合结构,然后进行了长达 μs 的模拟以完善结构。预测的CXCR4&配体结合结构与现有文献数据一致。根据Helix3-Helix6距离计算,带有Cys11-Cys40二硫键的hBD-3类似物能激活CXCR4,而其他类似物或突变体不能。这些结果有助于了解 CXCR4 受体与不同配体结合的动力学和激活机制。趋化因子受体 CXCR4 与癌症和多种疾病有关,但不同配体与该受体结合的分子细节仍不完整。在本文中,作者研究了不同形式的 CXCR4 与 CXCL12 和 hBD-3 的结合和相互作用,发现这两种配体都能与 CXCR4 在同一部位结合,而带有 Cys11-Cys40 二硫键的 hBD-3 类似物能激活 CXCR4。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Interaction and dynamics of chemokine receptor CXCR4 binding with CXCL12 and hBD-3
Chemokine receptor CXCR4 is involved in diverse diseases. A comparative study was conducted on CXCR4 embedded in a POPC lipid bilayer binding with CXCL12 in full and truncated forms, hBD-3 in wildtype, analog, and mutant forms based on in total 63 µs all-atom MD simulations. The initial binding structures of CXCR4 with ligands were predicted using HADDOCK docking or random-seed method, then μs-long simulations were performed to refine the structures. CXCR4&ligand binding structures predicted agree with available literature data. Both kinds of ligands bind stably to the N-terminus, extracellular loop 2 (ECL2), and ECL3 regions of CXCR4; the C2-C3 (K32-R38) region and occasionally the head of hBD-3 bind stably with CXCR4. hBD-3 analogs with Cys11-Cys40 disulfide bond can activate CXCR4 based on the Helix3-Helix6 distance calculation, but not other analogs or mutant. The results provide insight into understanding the dynamics and activation mechanism of CXCR4 receptor binding with different ligands. The chemokine receptor CXCR4 is involved in cancers and diverse diseases, however, molecular details surrounding the binding of different ligands to this receptor remain incomplete. Here, the authors study the binding and interaction between CXCR4 with CXCL12 and hBD-3 in different forms, and find that both ligands can bind with CXCR4 at the same site, and analogs of hBD-3 with a Cys11-Cys40 disulfide bond can activate CXCR4.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Communications Chemistry
Chemistry-General Chemistry
CiteScore
7.70
自引率
1.70%
发文量
146
审稿时长
13 weeks
期刊介绍:
Communications Chemistry is an open access journal from Nature Research publishing high-quality research, reviews and commentary in all areas of the chemical sciences. Research papers published by the journal represent significant advances bringing new chemical insight to a specialized area of research. We also aim to provide a community forum for issues of importance to all chemists, regardless of sub-discipline.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


