323.15 K 时混合硼酸盐体系 NaB(OH)4-Na2B4O5(OH)4-H2O 中水活度和固液平衡的同位素测定
IF 2
3区 工程技术
Q3 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
采用等压法研究了 323.15 K 下 NaB(OH)4-Na2B4O5(OH)4-H2O 三元体系的水活度和渗透系数。用等温溶解平衡法研究了 323.15 K 下 NaBO2-Na2B4O7-H2O 体系的溶解度和理化性质。该体系的相图由 NaBO2-4H2O 和 Na2B4O7-10H2O 两个单盐区、一个上述盐饱和不变点和两条单变量溶解度曲线组成。在皮策模型的基础上,用渗透系数和溶解度拟合了 Na2B4O5(OH)4 的皮策二元参数和混合参数。根据实验溶解度数据和参数计算了 Na2B4O7-10H2O 的溶解平衡常数。计算得出的 NaBO2-4H2O 和 Na2B4O7-10H2O 的溶解度和水活度、参数和溶解平衡常数与实验数据一致。本文章由计算机程序翻译,如有差异,请以英文原文为准。
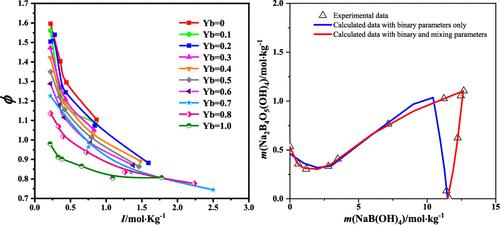
Isopiestic Determination of Water Activities and Solid–Liquid Equilibria in the Mixed Borate System NaB(OH)4–Na2B4O5(OH)4–H2O at 323.15 K
The water activities and osmotic coefficients in the ternary system NaB(OH)4–Na2B4O5(OH)4–H2O at 323.15 K were investigated with the isopiestic method. The solubilities and physicochemical properties in the NaBO2–Na2B4O7–H2O system at 323.15 K were studied with the isothermal dissolution equilibrium method. The phase diagram in the system consists of two single salt regions for NaBO2·4H2O and Na2B4O7·10H2O, one invariant point saturated with the above salts, and two univariant solubility curves. On the basis of the Pitzer model, the Pitzer binary parameters of Na2B4O5(OH)4 and mixing parameters were fitted with the osmotic coefficients and solubilities. The dissolution equilibrium constant of Na2B4O7·10H2O was calculated with the experimental solubility data and parameters. The calculated solubilities and water activities with the parameters and dissolution equilibrium constants of NaBO2·4H2O and Na2B4O7·10H2O agree with the experimental data.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Chemical & Engineering Data
工程技术-工程:化工
CiteScore
5.20
自引率
19.20%
发文量
324
审稿时长
2.2 months
期刊介绍:
The Journal of Chemical & Engineering Data is a monthly journal devoted to the publication of data obtained from both experiment and computation, which are viewed as complementary. It is the only American Chemical Society journal primarily concerned with articles containing data on the phase behavior and the physical, thermodynamic, and transport properties of well-defined materials, including complex mixtures of known compositions. While environmental and biological samples are of interest, their compositions must be known and reproducible. As a result, adsorption on natural product materials does not generally fit within the scope of Journal of Chemical & Engineering Data.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


