硅藻光收集复合物中的意外水通道:光收集与光保护之间转换的意义
IF 3.7
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
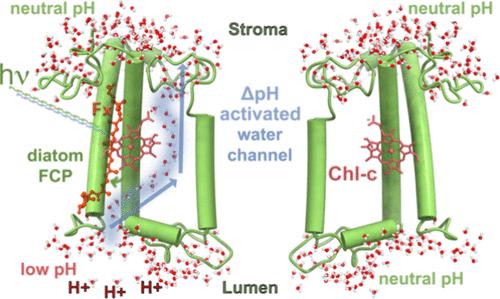
细胞中的许多重要过程都依赖于质子通过嵌入跨膜蛋白中的水丝进行转移。在这里,我们对硅藻的光收集复合物,即海洋硅藻三尖杉的岩藻黄素和叶绿素 a/c 结合蛋白(FCP)进行了超过 55 μs 的全原子模拟。硅藻是研究自然光合作用的独特模型,因为它们具有高效的光收集机制和强大的 pH 依赖性光保护机制。本研究报告了 FCP 单体、二聚体和四聚体在不同 pH 值下的动态变化。令人惊讶的是,我们发现在低 pH 值条件下,FCP 上有一条水通道,可选择性地使位于膜中央的 Chl-c2 色素的丙烯酸酯水合和质子化。质子动力学的 QM/MM 计算和定向 MD 模拟进一步证实了这些结果。结果表明,质子通过观察到的水通道在膜的腔侧和基质侧之间的跳跃事件是非常不利的。这种阻碍是由于靠近丙烯酸酯的 Arg31 和 Lys82 残基的存在,以及与水蒸发蛋白的水传导性非常相似的氢离子脱溶惩罚。此外,我们还提供了强有力的证据,证明这种已确定的水通道能够控制硅藻三棘藻中主要 FCP 复合物的光收集状态和光保护状态之间的转换。本文章由计算机程序翻译,如有差异,请以英文原文为准。
An Unexpected Water Channel in the Light-Harvesting Complex of a Diatom: Implications for the Switch between Light Harvesting and Photoprotection
Many important processes in cells depend on the transfer of protons through water wires embedded in transmembrane proteins. Herein, we have performed more than 55 μs all-atom simulations of the light-harvesting complex of a diatom, i.e., the fucoxanthin and chlorophyll a/c binding protein (FCP) from the marine diatom Phaeodactylum tricornutum. Diatoms are unique models to study natural photosynthesis as they exert an efficient light-harvesting machinery with a robust pH-dependent photoprotective mechanism. The present study reports on the dynamics of an FCP monomer, a dimer, and a tetramer at varying pH values. Surprisingly, we have identified at low pH a water channel across FCP that selectively hydrates and protonates the acrylate of a Chl-c2 pigment located in the middle of the membrane. These results are further supported by QM/MM calculations and steered MD simulations on the proton dynamics. It is shown that proton hopping events between the lumenal and stromal sides of the membrane through the observed water channel are highly disfavored. This hindrance is due to the presence of residues Arg31 and Lys82 close to the acrylate, along with an hydronium desolvation penalty that shows close similarities to the water conductance in aquaporins. Furthermore, we provide strong evidence that this identified water channel is governing the transition between light-harvesting and photoprotective states of the major FCP complex in the diatom P. tricornutum.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
3.70
自引率
0.00%
发文量
0
期刊介绍:
ACS Physical Chemistry Au is an open access journal which publishes original fundamental and applied research on all aspects of physical chemistry. The journal publishes new and original experimental computational and theoretical research of interest to physical chemists biophysical chemists chemical physicists physicists material scientists and engineers. An essential criterion for acceptance is that the manuscript provides new physical insight or develops new tools and methods of general interest. Some major topical areas include:Molecules Clusters and Aerosols; Biophysics Biomaterials Liquids and Soft Matter; Energy Materials and Catalysis

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


