工程化聚乳酸(PLGA)核脂壳混合纳米载体提高了伊立替康治疗结肠癌的疗效和安全性
IF 5.4
2区 医学
Q2 MATERIALS SCIENCE, BIOMATERIALS
引用次数: 0
摘要
聚乳酸-聚乙二醇(PLGA)是一种生物相容性和可生物降解的共聚物,在生物医学应用中获得了很高的认可度。本研究采用绿色工艺,在生物相容性锌脯氨酸引发剂存在下,通过开环聚合合成了 PLGA(Mw = 13,900)。以 1,2-二硬脂酰-sn-甘油-3-膦酰乙醇胺和 1.2-二硬脂酰-sn-甘油-3-膦酰乙醇胺配制了负载高效 PLGA 内核-脂质外壳杂化纳米载体(脂质体,LPs)的伊立替康(Ir)、采用纳米沉淀法,以大豆卵磷脂为原料,配制了 1,2-二硬脂酰-sn-甘油-3-磷脂酰乙醇胺和 1,2-二硬脂酰-sn-甘油-3-磷脂酰乙醇胺-N-[氨基(聚乙二醇)-2000](DSPE-PEG-2000),并分别命名为 P-DSPE-Ir 和 P-DSPE-PEG-Ir。实验进一步验证了所配制的 LPs 在结肠癌应用中的理化性质和生物学潜力。研究了一种水溶性较差的化疗药物(Ir)在结肠癌治疗中的潜在输送能力。成功制备了具有可控尺寸(80-120 nm)和表面电荷(∼ -35 mV)的 LPs,并研究了其持续释放特性和对 CT-26 结肠癌细胞的细胞毒性。在 CT-26 异种移植瘤携带 Balb/C 小鼠体内进行的生物分布和肿瘤部位保留研究表明,该药物可被肿瘤吸收并长时间保留。与 P-DSPE-Ir 不同的是,P-DSPE-PEG-Ir LP 在 CT-26(皮下肿瘤模型)中与未处理组和空白制剂处理组相比,在 10 毫克伊立替康/千克剂量的 4 个疗程后表现出明显的肿瘤生长延迟。对优化配方进行的急性毒性研究证实了 LP 的生物相容性和安全性。总之,这项概念验证研究表明,基于PLGA的LPs提高了伊立替康的疗效和生物利用度,减少了中性粒细胞减少症,可用于抗击结肠癌。本文章由计算机程序翻译,如有差异,请以英文原文为准。
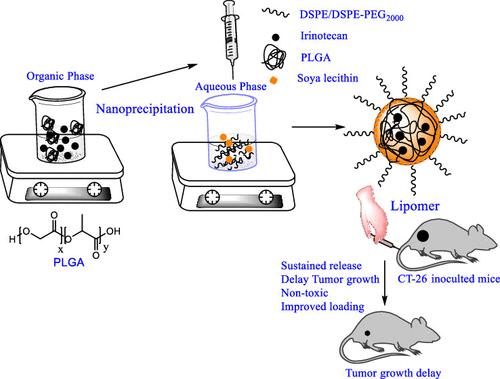
Engineered PLGA Core–Lipid Shell Hybrid Nanocarriers Improve the Efficacy and Safety of Irinotecan to Combat Colon Cancer
Poly(lactide-co-glycolide) (PLGA) is a biocompatible and biodegradable copolymer that has gained high acceptance in biomedical applications. In the present study, PLGA (Mw = 13,900) was synthesized by ring-opening polymerization in the presence of a biocompatible zinc–proline initiator through a green route. Irinotecan (Ir) loaded with efficient PLGA core–lipid shell hybrid nanocarriers (lipomers, LPs) were formulated with 1,2-distearoyl-sn-glycero-3-phosphoethanolamine and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino (polyethylene glycol)-2000] (DSPE-PEG-2000), using soya lecithin, by a nanoprecipitation method, and the fabricated LPs were designated as P-DSPE-Ir and P-DSPE-PEG-Ir, respectively. The formulated LPs were further validated for their physicochemical properties and biological potential for colon cancer application. The potential delivery of a poorly water-soluble chemotherapeutic drug (Ir) was studied for the treatment of colon cancer. LPs were successfully prepared, providing controlled size (80–120 nm) and surface charge (∼ −35 mV), and the sustained release properties and cytotoxicity against CT-26 colon cancer cells were studied. The in vivo biodistribution and tumor site retention in CT-26 xenograft tumor-bearing Balb/C mice showed promising results for tumor uptake and retention for a prolonged time period. Unlike P-DSPE-Ir, the P-DSPE-PEG-Ir LP exhibited significant tumor growth delay as compared to untreated and blank formulation-treated groups in CT-26 (subcutaneous tumor model) after 4 treatments of 10 mg irinotecan/kg dose. The biocompatibility and safety of the LPs were confirmed by an acute toxicity study of the optimized formulation. Overall, this proof-of-concept study demonstrates that the PLGA-based LPs improve the efficacy and bioavailability and decrease neutropenia of Ir to combat colon cancer.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Biomaterials Science & Engineering
Materials Science-Biomaterials
CiteScore
10.30
自引率
3.40%
发文量
413
期刊介绍:
ACS Biomaterials Science & Engineering is the leading journal in the field of biomaterials, serving as an international forum for publishing cutting-edge research and innovative ideas on a broad range of topics:
Applications and Health – implantable tissues and devices, prosthesis, health risks, toxicology
Bio-interactions and Bio-compatibility – material-biology interactions, chemical/morphological/structural communication, mechanobiology, signaling and biological responses, immuno-engineering, calcification, coatings, corrosion and degradation of biomaterials and devices, biophysical regulation of cell functions
Characterization, Synthesis, and Modification – new biomaterials, bioinspired and biomimetic approaches to biomaterials, exploiting structural hierarchy and architectural control, combinatorial strategies for biomaterials discovery, genetic biomaterials design, synthetic biology, new composite systems, bionics, polymer synthesis
Controlled Release and Delivery Systems – biomaterial-based drug and gene delivery, bio-responsive delivery of regulatory molecules, pharmaceutical engineering
Healthcare Advances – clinical translation, regulatory issues, patient safety, emerging trends
Imaging and Diagnostics – imaging agents and probes, theranostics, biosensors, monitoring
Manufacturing and Technology – 3D printing, inks, organ-on-a-chip, bioreactor/perfusion systems, microdevices, BioMEMS, optics and electronics interfaces with biomaterials, systems integration
Modeling and Informatics Tools – scaling methods to guide biomaterial design, predictive algorithms for structure-function, biomechanics, integrating bioinformatics with biomaterials discovery, metabolomics in the context of biomaterials
Tissue Engineering and Regenerative Medicine – basic and applied studies, cell therapies, scaffolds, vascularization, bioartificial organs, transplantation and functionality, cellular agriculture
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


