用于可充电镁电池的 Cu3VSe4 阴极:诱导互锁和置换反应的有利化学和电子结构
IF 18.5
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
可充电镁电池是一种成本低、安全性高的优势储能技术,但高性能正极材料的设计是目前的一大难点。本文综合考虑化学结构和电子结构,制备了一种新型 Cu3VSe4 阴极材料。中间带半导体 Cu3VSe4 具有包含交错三维隧道的立方晶体结构。V 原子和 Se 原子形成高共价比例的化学键,并通过 V-Se 键促进电荷的分散。由于这些特点,Cu3VSe4 在铜、钒和硒元素共同氧化还原的情况下可提供 251 mAh g-1 的高容量,并且在 15 A g-1 的条件下具有 44 mAh g-1 的出色速率性能。更突出的是,在实现 3.0 mg cm-2 的高负载质量时,速率能力没有明显衰减,这非常有利于在实际应用中与高容量镁金属阳极配对。机理研究和理论计算表明,Cu3VSe4 首先发生镁插层反应,然后发生置换反应,在置换反应过程中晶体结构得以保持,从而提高了反应的可逆性和循环稳定性。这些发现揭示了基于化学和电子结构综合考虑的可充电镁电池阴极的合理设计原理。本文章由计算机程序翻译,如有差异,请以英文原文为准。
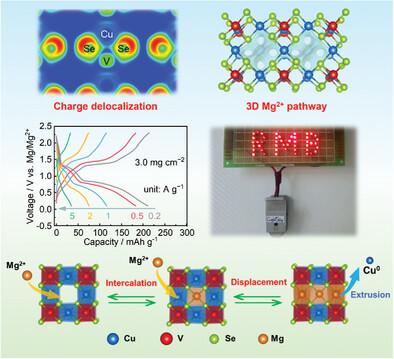
Cu3VSe4 Cathode for Rechargeable Magnesium Batteries: Favorable Chemical and Electronic Structures Inducing Intercalation and Displacement Reactions
Rechargeable Mg batteries are an advantageous energy-storage technology with low cost and high safety, but the design of high-performance cathode materials is currently the major difficulty. Herein, a new cathode material of Cu3VSe4 is fabricated with a comprehensive consideration of the chemical and electronic structures. The intermediate band semiconductor Cu3VSe4 has a cubic crystal structure containing interlaced 3D tunnels. The V and Se atoms form chemical bonds with high covalent proportions and facilitate the charge delocalization via the V‒Se bonds. Because of these features, Cu3VSe4 provides a high capacity of 251 mAh g‒1 with co-redox of Cu, V, and Se elements and an outstanding rate performance of 44 mAh g‒1 at 15 A g‒1. Prominently, a high mass load of 3.0 mg cm‒2 is achieved without obvious rate capability decay, which is quite favorable to pair with the high-capacity Mg metal anode in practical application. The mechanism investigation and theoretical computation demonstrate that Cu3VSe4 undergoes first a Mg-intercalation and then a displacement reaction, during which the crystal structure is maintained, assisting the reaction reversibility and cycling stability. These findings reveal a rational design principle of rechargeable Mg battery cathodes based on a comprehensive consideration of chemical and electronic structures.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Advanced Functional Materials
工程技术-材料科学:综合
CiteScore
29.50
自引率
4.20%
发文量
2086
审稿时长
2.1 months
期刊介绍:
Firmly established as a top-tier materials science journal, Advanced Functional Materials reports breakthrough research in all aspects of materials science, including nanotechnology, chemistry, physics, and biology every week.
Advanced Functional Materials is known for its rapid and fair peer review, quality content, and high impact, making it the first choice of the international materials science community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


