扰乱 TET2 凝聚可促进全基因组 DNA 甲基化异常并抑制白血病细胞生长
IF 17.3
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
十-十一易位(TET)二氧合酶家族在细胞分裂和品系分化过程中维持稳定的局部 DNA 去甲基化。作为 TET 酶的主要催化产物,5-羟甲基胞嘧啶以组织依赖的方式选择性地富集在特定的基因组区域,如增强子。然而,这种选择性的内在机制仍未解决。在这里,我们揭示了 TET2 中的一个低复杂性插入结构域,它能促进 TET2 与表观遗传调节剂(如 UTX 和 MLL4)的生物分子缩聚。这种共同凝结为 DNA 的精确去甲基化创造了有利的染色质环境。破坏低复杂性插入介导的缩聚会改变 TET2 的基因组结合,从而导致杂乱的 DNA 去甲基化和基因组重组。这些变化会影响与白血病发生有关的关键基因的表达,从而抑制白血病细胞的增殖。总之,这项研究证实了 TET2 冷凝在协调精确的 DNA 去甲基化和基因转录以支持肿瘤细胞生长方面的关键作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
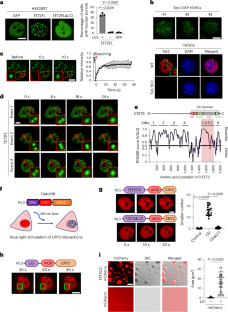

Perturbing TET2 condensation promotes aberrant genome-wide DNA methylation and curtails leukaemia cell growth
The ten-eleven translocation (TET) family of dioxygenases maintain stable local DNA demethylation during cell division and lineage specification. As the major catalytic product of TET enzymes, 5-hydroxymethylcytosine is selectively enriched at specific genomic regions, such as enhancers, in a tissue-dependent manner. However, the mechanisms underlying this selectivity remain unresolved. Here we unveil a low-complexity insert domain within TET2 that facilitates its biomolecular condensation with epigenetic modulators, such as UTX and MLL4. This co-condensation fosters a permissive chromatin environment for precise DNA demethylation. Disrupting low-complexity insert-mediated condensation alters the genomic binding of TET2 to cause promiscuous DNA demethylation and genome reorganization. These changes influence the expression of key genes implicated in leukaemogenesis to curtail leukaemia cell proliferation. Collectively, this study establishes the pivotal role of TET2 condensation in orchestrating precise DNA demethylation and gene transcription to support tumour cell growth. Guo, Hong et al. report that TET2 condensation maintains proper DNA demethylation at specific genomic loci, which can be targeted to alter gene expression and impair leukaemia growth.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Nature Cell Biology
生物-细胞生物学
CiteScore
28.40
自引率
0.90%
发文量
219
审稿时长
3 months
期刊介绍:
Nature Cell Biology, a prestigious journal, upholds a commitment to publishing papers of the highest quality across all areas of cell biology, with a particular focus on elucidating mechanisms underlying fundamental cell biological processes. The journal's broad scope encompasses various areas of interest, including but not limited to:
-Autophagy
-Cancer biology
-Cell adhesion and migration
-Cell cycle and growth
-Cell death
-Chromatin and epigenetics
-Cytoskeletal dynamics
-Developmental biology
-DNA replication and repair
-Mechanisms of human disease
-Mechanobiology
-Membrane traffic and dynamics
-Metabolism
-Nuclear organization and dynamics
-Organelle biology
-Proteolysis and quality control
-RNA biology
-Signal transduction
-Stem cell biology
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


