桑蚜 Anomoneura mori 中双重细菌共生体基因组的高度还原互补性。
摘要
必须受宿主限制的细菌的基因组会发生轻微的有害突变,导致体积明显缩小。车轴虫(半翅目)是一种吸食韧皮部汁液的昆虫,它有一个被称为细菌组的特殊器官,通常携带两种垂直传播的细菌共生体:主要共生体 "Candidatus Carsonella ruddii"(伽马蛋白杆菌)和次要共生体,后者在车轴虫品系中具有系统发育上的多样性。研究发现,几个卡森氏菌系的基因组明显缩小(158-174 kb),富含 AT(14.0-17.9% GC),结构保守,具有类似的基因库,专门用于合成韧皮部汁液中稀缺的必需氨基酸。然而,目前关于次生共生体的基因组信息非常有限。因此,本研究调查了桑树蚜虫 Anomoneura mori(Psyllidae)中与细菌体相关的双重共生体 Secondary_AM(伽马蛋白杆菌)和 Carsonella_AM 的基因组。研究结果表明,Secondary_AM 基因组与 Carsonella 菌系(包括 Carsonella_AM(169 120 bp,16.2% GC))一样小且富含 AT(229 822 bp,17.3% GC),这意味着 Secondary_AM 与 Carsonella 一样是一种进化古老的强制性互生菌。系统发生组分析表明,Secondary_AM 与最近报道的 Cacopsylla spp.(Psyllidae)的 "Candidatus Psyllophila symbiotica"(221-237 kb,17.3-18.6% GC)基因组是姊妹。Secondary_AM和Psyllophila基因组显示出高度保守的同源性,共享所有用于补充Carsonella不完全色氨酸生物合成途径的基因和合成B族维生素的基因。然而,硫同化基因和类胡萝卜素合成基因分别只在 Secondary_AM 和车前子中保留下来,这表明基因正在发生沉默。平均核苷酸同一性、基因直向同源物相似性、全基因组同源性和替换率表明,Secondary_AM/Psyllophila 基因组比 Carsonella 基因组更易变异。
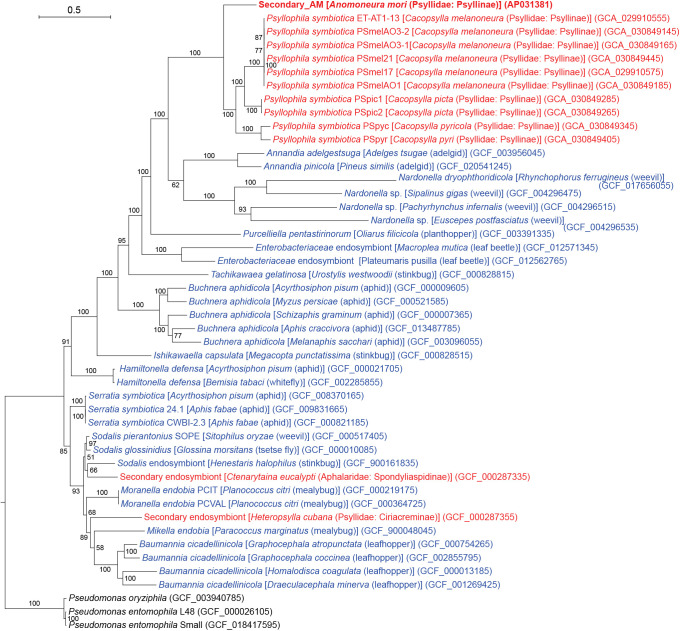
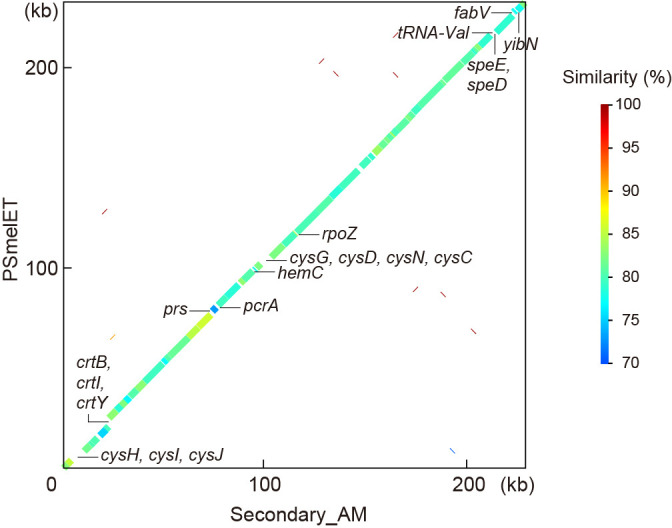
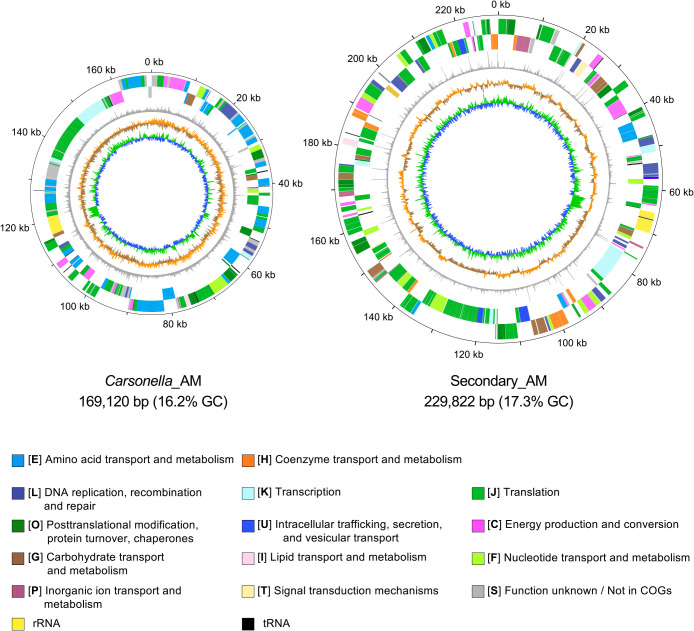
The genomes of obligately host-restricted bacteria suffer from accumulating mildly deleterious mutations, resulting in marked size reductions. Psyllids (Hemiptera) are phloem sap-sucking insects with a specialized organ called the bacteriome, which typically harbors two vertically transmitted bacterial symbionts: the primary symbiont "Candidatus Carsonella ruddii" (Gammaproteobacteria) and a secondary symbiont that is phylogenetically diverse among psyllid lineages. The genomes of several Carsonella lineages were revealed to be markedly reduced (158-174 kb), AT-rich (14.0-17.9% GC), and structurally conserved with similar gene inventories devoted to synthesizing essential amino acids that are scarce in the phloem sap. However, limited genomic information is currently available on secondary symbionts. Therefore, the present study investigated the genomes of the bacteriome-associated dual symbionts, Secondary_AM (Gammaproteobacteria) and Carsonella_AM, in the mulberry psyllid Anomoneura mori (Psyllidae). The results obtained revealed that the Secondary_AM genome is as small and AT-rich (229,822 bp, 17.3% GC) as those of Carsonella lineages, including Carsonella_AM (169,120 bp, 16.2% GC), implying that Secondary_AM is an evolutionarily ancient obligate mutualist, as is Carsonella. Phylogenomic ana-lyses showed that Secondary_AM is sister to "Candidatus Psyllophila symbiotica" of Cacopsylla spp. (Psyllidae), the genomes of which were recently reported (221-237 kb, 17.3-18.6% GC). The Secondary_AM and Psyllophila genomes showed highly conserved synteny, sharing all genes for complementing the incomplete tryptophan biosynthetic pathway of Carsonella and those for synthesizing B vitamins. However, sulfur assimilation and carotenoid-synthesizing genes were only retained in Secondary_AM and Psyllophila, respectively, indicating ongoing gene silencing. Average nucleotide identity, gene ortholog similarity, genome-wide synteny, and substitution rates suggest that the Secondary_AM/Psyllophila genomes are more labile than Carsonella genomes.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


