镁介导的芳基 2-吡啶酯与芳基溴化物的交叉亲电偶联,通过原位形成的芳基镁中间体合成酮。
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
The Journal of Organic Chemistry
Pub Date : 2024-09-20
Epub Date: 2024-09-09
DOI:10.1021/acs.joc.4c01851
引用次数: 0
摘要
在没有过渡金属催化剂的情况下,芳基 2-吡啶酯可以在镁作为还原金属的帮助下与芳基溴化物高效地发生交电偶联反应,从而得到不对称的二芳基酮,产率适中甚至很高,并且具有广泛的官能度兼容性。此外,该反应易于放大,可用于生物活性分子的后期修饰。初步的机理研究表明,该偶联反应可能是通过原位形成芳基镁试剂作为关键中间体进行的。本文章由计算机程序翻译,如有差异,请以英文原文为准。
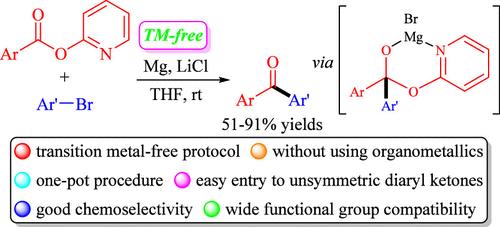
Magnesium-Mediated Cross-Electrophile Couplings of Aryl 2-Pyridyl Esters with Aryl Bromides for Ketone Synthesis through In Situ-Formed Arylmagnesium Intermediates.
Aryl 2-pyridyl esters could efficiently undergo cross-electrophile couplings with aryl bromides with the aid of magnesium as a reducing metal in the absence of a transition-metal catalyst, leading to the unsymmetrical diaryl ketones in modest to good yields with wide functionality compatibility. In addition, the reaction could be easily scaled up and applied in the late-stage modification of biologically active molecules. Preliminary mechanistic study showed that the coupling reaction presumably proceeds through the in situ formation of arylmagnesium reagents as key intermediates.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


