概述:arborisidine 和 arbornamine 的全合成。
IF 3.9
2区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
摘要
Arborisidine 和 Arbornamine 是从马来亚 Kopsia arborea 植物中分离出的两种单萜吲哚生物碱。本综述提供了有关这些生物碱的全合成和正式合成的宝贵信息。本综述中讨论的合成策略,如 Pictet-Spengler 环化、化学和立体选择性氧化环化、迈克尔/曼尼希级联过程和分子内 N-烷基化,可用于开发合成这些化合物和其他类似化合物的新方法。本文章由计算机程序翻译,如有差异,请以英文原文为准。
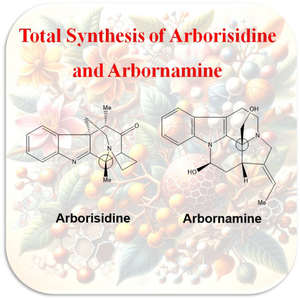
An overview: total synthesis of arborisidine, and arbornamine.
Arborisidine and Arbornamine are two monoterpenoid indole alkaloids that were isolated from the Malayan Kopsia arborea plant. This review provides valuable information about the total and formal syntheses of these alkaloids. The synthesis strategies discussed in this review, such as Pictet-Spengler cyclization, chemo- and stereoselective oxidative cyclization, Michael/Mannich cascade process, and intramolecular N-alkylation, can be useful for developing new methods to synthesize these and other similar compounds.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Molecular Diversity
化学-化学综合
CiteScore
7.30
自引率
7.90%
发文量
219
审稿时长
2.7 months
期刊介绍:
Molecular Diversity is a new publication forum for the rapid publication of refereed papers dedicated to describing the development, application and theory of molecular diversity and combinatorial chemistry in basic and applied research and drug discovery. The journal publishes both short and full papers, perspectives, news and reviews dealing with all aspects of the generation of molecular diversity, application of diversity for screening against alternative targets of all types (biological, biophysical, technological), analysis of results obtained and their application in various scientific disciplines/approaches including:
combinatorial chemistry and parallel synthesis;
small molecule libraries;
microwave synthesis;
flow synthesis;
fluorous synthesis;
diversity oriented synthesis (DOS);
nanoreactors;
click chemistry;
multiplex technologies;
fragment- and ligand-based design;
structure/function/SAR;
computational chemistry and molecular design;
chemoinformatics;
screening techniques and screening interfaces;
analytical and purification methods;
robotics, automation and miniaturization;
targeted libraries;
display libraries;
peptides and peptoids;
proteins;
oligonucleotides;
carbohydrates;
natural diversity;
new methods of library formulation and deconvolution;
directed evolution, origin of life and recombination;
search techniques, landscapes, random chemistry and more;
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


