ATG2A-ATG9A 复合物脂质转移的结构基础
IF 12.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
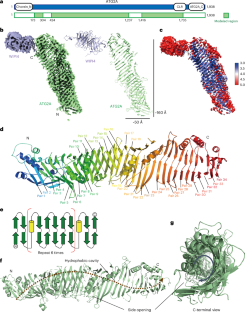
自噬的特点是形成称为自噬体的双膜囊泡。自噬相关蛋白(ATGs)2A和9A在自噬过程中起着至关重要的作用,它们介导自噬体形成过程中膜间的脂质转移和再平衡。在此,我们报告了人 ATG2A 与 WD 重覆蛋白与磷酸肌酸 4(WIPI4)相互作用复合物在 3.2 Å 的冷冻电镜结构,以及 ATG2A-WIPI4-ATG9A 复合物在 7 Å 全局分辨率下的结构。在分子动力学模拟的基础上,我们提出了从供体膜中提取脂质的机制。我们的分析发现 ATG9A-ATG2A 复合物的配比为 3:1,ATG9A 侧孔与 ATG2A 脂质转移腔直接对齐,ATG9A 三聚体与杆状 ATG2A 的 N 端和 C 端都有相互作用。ATG2A 脂质体结合状态的低温电子断层扫描显示,ATG2A 以不同的方向拴住脂质囊泡。总之,这项研究为吞噬膜的生长提供了分子基础,并从结构上揭示了自噬体形成过程中空间耦合脂质运输和再平衡的过程。本文章由计算机程序翻译,如有差异,请以英文原文为准。

Structural basis for lipid transfer by the ATG2A–ATG9A complex
Autophagy is characterized by the formation of double-membrane vesicles called autophagosomes. Autophagy-related proteins (ATGs) 2A and 9A have an essential role in autophagy by mediating lipid transfer and re-equilibration between membranes for autophagosome formation. Here we report the cryo-electron microscopy structures of human ATG2A in complex with WD-repeat protein interacting with phosphoinositides 4 (WIPI4) at 3.2 Å and the ATG2A–WIPI4–ATG9A complex at 7 Å global resolution. On the basis of molecular dynamics simulations, we propose a mechanism of lipid extraction from the donor membranes. Our analysis revealed 3:1 stoichiometry of the ATG9A–ATG2A complex, directly aligning the ATG9A lateral pore with ATG2A lipid transfer cavity, and an interaction of the ATG9A trimer with both the N-terminal and the C-terminal tip of rod-shaped ATG2A. Cryo-electron tomography of ATG2A liposome-binding states showed that ATG2A tethers lipid vesicles at different orientations. In summary, this study provides a molecular basis for the growth of the phagophore membrane and lends structural insights into spatially coupled lipid transport and re-equilibration during autophagosome formation. The study presents cryo-electron microscopy structures of human autophagy-related protein (ATG) 2A in complex with WD-repeat protein interacting with phosphoinositides 4 (WIPI4) and the ATG2A–WIPI4–ATG9A complex, providing insights into spatially coupled lipid transport and re-equilibration during autophagosome formation.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


