结合迈耶-舒斯特重排和不对称烯生物还原,从原炔醇制备β,β-二取代烷-2-酮
IF 4.4
2区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
摘要
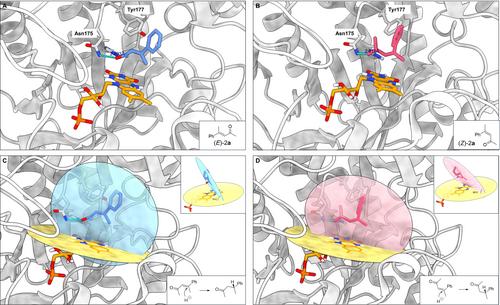
金(I)N-杂环碳络合物与烯还原酶(ERED)的结合,使对映体纯β,β-二取代酮的合成成为可能。该方法包括使用[1,3-双(2,6-二异丙基苯基)咪唑-2-亚基]-[双(三氟甲磺酰基)-亚胺]金(I)(IPrAuNTf2)对外消旋丙炔基叔醇进行迈耶-舒斯特重排、然后使用 Zymomonas mobilis ERED(NCR-ERED)对α,β-不饱和酮中间体进行不对称烯还原。该化学酶级联使用模型底物进行了优化,其中 E/Z 异构体都生成了 (R)- 酮,并使用硅分子对接实验对其进行了合理化。级联反应随后被直接用于生产一系列对映体纯的 (R)-4 取代-2-酮。本文章由计算机程序翻译,如有差异,请以英文原文为准。
β,β‐Disubstituted Alkan‐2‐ones from Propargylic Alcohols Combining a Meyer‐Schuster Rearrangement and Asymmetric Alkene Bioreduction
The combination of a gold(I) N‐heterocyclic carbene complex and an ene‐reductase (ERED) has made possible the synthesis of enantiopure β,β‐disubstituted ketones in a one‐pot concurrent approach. The protocol consists of the Meyer‐Schuster rearrangement of racemic propargylic tertiary alcohols using [1,3‐bis(2,6‐diisopropylphenyl)imidazol‐2‐ylidene]‐[bis(trifluoromethanesulfonyl)‐imide]gold(I) (IPrAuNTf2), followed by an asymmetric alkene reduction of the α,β‐unsaturated ketone intermediate using the Zymomonas mobilis ERED (NCR‐ERED). The chemoenzymatic cascade was optimised with a model substrate, where E/Z‐isomers both generated the (R)‐ketone, which was rationalised using in silico molecular docking experiments. The cascade was then applied towards the production of a series of (R)‐4‐substituted‐alkan‐2‐ones in enantiopure form in a straightforward manner.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Advanced Synthesis & Catalysis
化学-应用化学
CiteScore
9.40
自引率
7.40%
发文量
447
审稿时长
1.8 months
期刊介绍:
Advanced Synthesis & Catalysis (ASC) is the leading primary journal in organic, organometallic, and applied chemistry.
The high impact of ASC can be attributed to the unique focus of the journal, which publishes exciting new results from academic and industrial labs on efficient, practical, and environmentally friendly organic synthesis. While homogeneous, heterogeneous, organic, and enzyme catalysis are key technologies to achieve green synthesis, significant contributions to the same goal by synthesis design, reaction techniques, flow chemistry, and continuous processing, multiphase catalysis, green solvents, catalyst immobilization, and recycling, separation science, and process development are also featured in ASC. The Aims and Scope can be found in the Notice to Authors or on the first page of the table of contents in every issue.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


