在固体电解质反应器中通过阳离子穿梭进行硝酸盐还原成氨的电化学反应
IF 42.8
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
虽然电化学硝酸盐还原成氨是水处理和合成氨的一条很有前景的途径,但该领域的一个关键挑战是这种电化学系统需要高浓度的支撑电解质。在此,我们报告了一种三室多孔固体电解质反应器设计,该设计与阳离子屏蔽效应相结合,可在无支撑电解质的情况下进行高效硝酸盐还原反应。通过将处理过的水从阴极室送入中间多孔固体电解质层,我们可以实现碱金属阳离子从中间层穿梭回阴极室的循环,从而提高硝酸盐还原选择性并抑制氢演化副反应。该反应器系统可在典型废水硝酸盐浓度为 2,000 ppm 的情况下,以实用的电流密度(100 mA cm-2)提供较高的氨法拉第效率(90%),从而无需电解质回收过程即可获得高纯度的出水和 NH3(g)产品。本文章由计算机程序翻译,如有差异,请以英文原文为准。
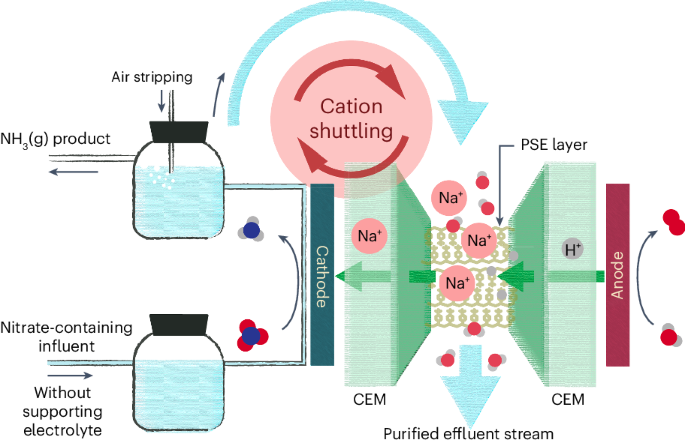
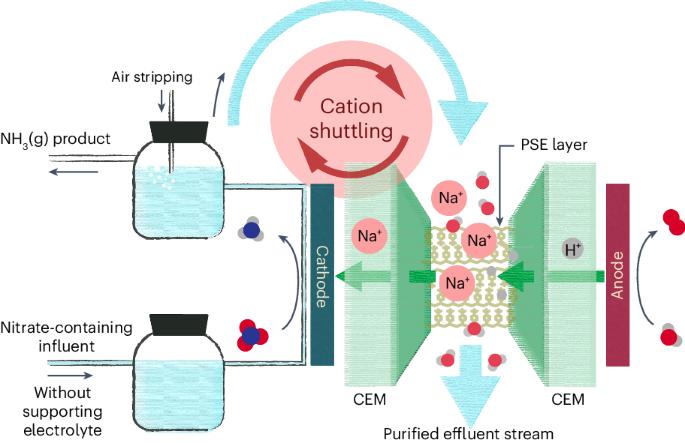
Electrochemical nitrate reduction to ammonia with cation shuttling in a solid electrolyte reactor
While electrochemical nitrate reduction to ammonia represents a promising route for water treatment and ammonia generation, one critical challenge in the field is the need for high-concentration supporting electrolytes in this electrochemical system. Here we report a three-chamber porous solid electrolyte reactor design coupled with cation shielding effects for efficient nitrate reduction reaction without supporting electrolytes. By feeding treated water from the cathode chamber to the middle porous solid electrolyte layer, we can realize an alkali metal cation shuttling loop from the middle layer back into the cathode chamber to boost the nitrate reduction selectivity and suppress the hydrogen evolution side reaction. This reactor system can deliver high ammonia Faradaic efficiencies (>90%) at practical current densities (>100 mA cm−2) under a typical wastewater nitrate concentration of 2,000 ppm, enabling a high-purity water effluent and NH3(g) as products with no need for electrolyte recovery processes. Electrocatalysis offers a route to improving the treatment of wastewater, yet the need for supporting electrolytes complicates the purification of products. Here a cell is designed based on a porous solid electrolyte layer with a cation shuttling strategy that allows direct conversion of nitrate-containing wastewater into NH3(g) and purified water.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Catalysis
Chemical Engineering-Bioengineering
CiteScore
52.10
自引率
1.10%
发文量
140
期刊介绍:
Nature Catalysis serves as a platform for researchers across chemistry and related fields, focusing on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, encompassing both fundamental and applied studies. With a particular emphasis on advancing sustainable industries and processes, the journal provides comprehensive coverage of catalysis research, appealing to scientists, engineers, and researchers in academia and industry.
Maintaining the high standards of the Nature brand, Nature Catalysis boasts a dedicated team of professional editors, rigorous peer-review processes, and swift publication times, ensuring editorial independence and quality. The journal publishes work spanning heterogeneous catalysis, homogeneous catalysis, and biocatalysis, covering areas such as catalytic synthesis, mechanisms, characterization, computational studies, nanoparticle catalysis, electrocatalysis, photocatalysis, environmental catalysis, asymmetric catalysis, and various forms of organocatalysis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


