合成化学中的多任务卤代烃
IF 1.5
4区 化学
Q3 CHEMISTRY, ORGANIC
引用次数: 0
摘要
卤代炔类化合物可以从多种前体中获得,在合成化学中作为炔化和/或卤化试剂用途广泛,能够以选择性的方式简便而多样地传递功能分子。最近,卤代炔类化合物的反应性和底物范围也得到了进一步的发展,从而促进了融合杂环的快速传递。本文章由计算机程序翻译,如有差异,请以英文原文为准。
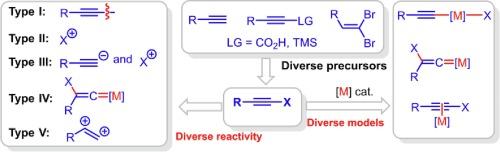
Multitasking haloalkynes in synthetic chemistry
Haloalkynes that could be accessed from diverse precursors, are versatile in synthetic chemistry as alkynylation and/or halogenation reagents, enabling concise and divergent delivery of functional molecules in selective manners. Recently, additional reactivity and substrate scope of haloalkynes, was also developed, facilitating expedient delivery of fused heterocycles.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Tetrahedron Letters
化学-有机化学
CiteScore
3.50
自引率
5.60%
发文量
521
审稿时长
28 days
期刊介绍:
Tetrahedron Letters provides maximum dissemination of outstanding developments in organic chemistry. The journal is published weekly and covers developments in techniques, structures, methods and conclusions in experimental and theoretical organic chemistry. Rapid publication of timely and significant research results enables researchers from all over the world to transmit quickly their new contributions to large, international audiences.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


