肝细胞特异性黑色素皮质素 1 受体缺失会扰乱脂肪酸代谢并促进脂肪细胞肥大
IF 4.2
2区 医学
Q1 ENDOCRINOLOGY & METABOLISM
引用次数: 0
摘要
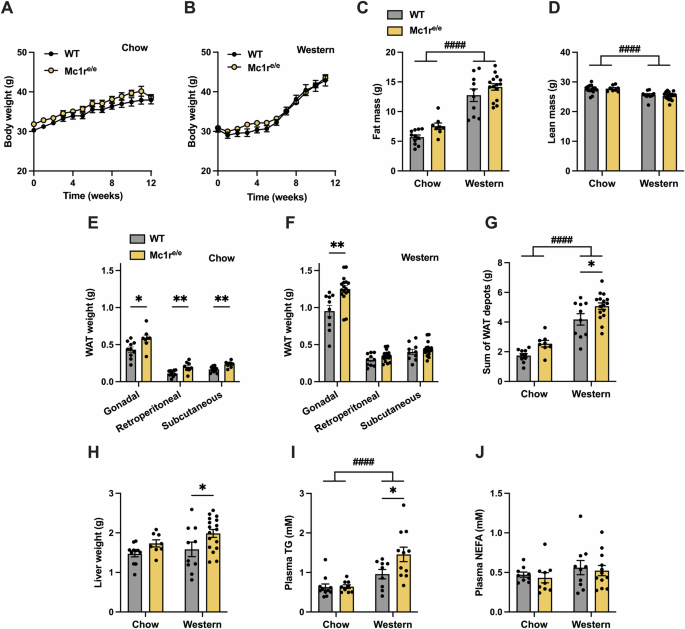
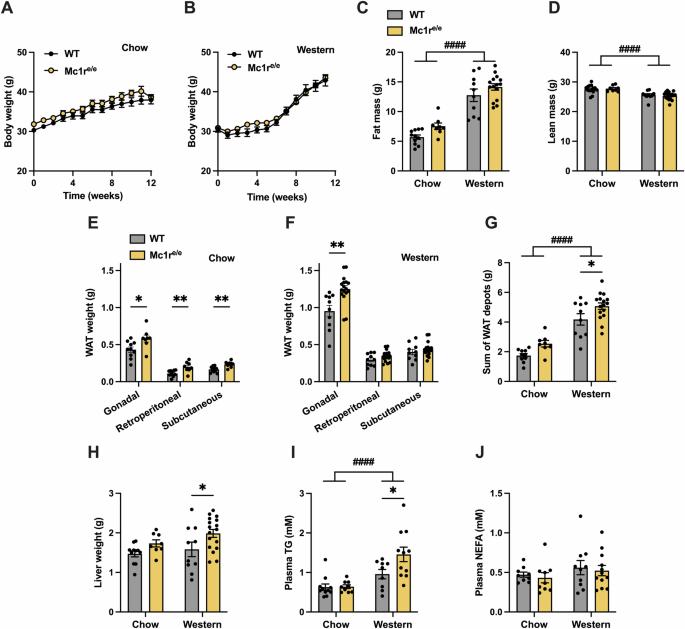
背景/目的:黑色素皮质素通过五种不同的黑色素皮质素受体(MC1R - MC5R)介导其生物功能。MC1R 在皮肤和白细胞中表达,调节皮肤色素沉着和炎症反应。MC1R 也存在于肝脏和白色脂肪组织中,但其在这些组织中的功能作用尚不清楚。本研究旨在确定 MC1R 在脂肪酸代谢中的调节作用:方法:雄性隐性黄色(Mc1re/e)小鼠(全球 MC1R 缺乏模型)和雄性肝细胞特异性 MC1R 缺乏小鼠(Mc1r LKO)均以饲料或西式饮食喂养 12 周。这些小鼠模型的特征包括体重和组成、肝脏脂肪含量、脂肪组织质量和形态、葡萄糖代谢和脂质代谢。此外,还使用 qPCR 和 RNA 测序分析来研究肝脏和脂肪组织的基因表达谱。用 HepG2 细胞和小鼠原代肝细胞研究药理激活 MC1R 的效果:结果:与野生型小鼠相比,周粮和西式饮食喂养的Mc1re/e小鼠的肝脏重量、白色脂肪组织质量和血浆甘油三酯(TG)浓度均有所增加。出现这种表型时,食物摄入量、体重、体力活动或葡萄糖代谢均无明显变化。Mc1r LKO 小鼠也表现出类似的表型,其特点是脂肪囤积增多、脂肪细胞肥大以及肝脏和血浆中甘油三酯的积累增加。在基因表达方面,Mc1r LKO 小鼠肝脏中新生脂肪生成、炎症和细胞凋亡的标志物上调,而这些小鼠白色脂肪组织中调节脂肪分解的酶下调。在培养的肝细胞中,MC1R的选择性激活降低了ChREBP的表达,而ChREBP是脂肪生成的核心转录因子:结论:肝细胞特异性缺失 MC1R 会扰乱肝脏中的脂肪酸代谢,导致以脂肪细胞肥大和肝脏及血液循环中 TG 累积增强为特征的肥胖表型。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Hepatocyte-specific loss of melanocortin 1 receptor disturbs fatty acid metabolism and promotes adipocyte hypertrophy
Melanocortins mediate their biological functions via five different melanocortin receptors (MC1R - MC5R). MC1R is expressed in the skin and leukocytes, where it regulates skin pigmentation and inflammatory responses. MC1R is also present in the liver and white adipose tissue, but its functional role in these tissues is unclear. This study aimed at determining the regulatory role of MC1R in fatty acid metabolism. Male recessive yellow (Mc1re/e) mice, a model of global MC1R deficiency, and male hepatocyte-specific MC1R deficient mice (Mc1r LKO) were fed a chow or Western diet for 12 weeks. The mouse models were characterized for body weight and composition, liver adiposity, adipose tissue mass and morphology, glucose metabolism and lipid metabolism. Furthermore, qPCR and RNA sequencing analyses were used to investigate gene expression profiles in the liver and adipose tissue. HepG2 cells and primary mouse hepatocytes were used to study the effects of pharmacological MC1R activation. Chow- and Western diet-fed Mc1re/e showed increased liver weight, white adipose tissue mass and plasma triglyceride (TG) concentration compared to wild type mice. This phenotype occurred without significant changes in food intake, body weight, physical activity or glucose metabolism. Mc1r LKO mice displayed a similar phenotype characterized by larger fat depots, increased adipocyte hypertrophy and enhanced accumulation of TG in the liver and plasma. In terms of gene expression, markers of de novo lipogenesis, inflammation and apoptosis were upregulated in the liver of Mc1r LKO mice, while enzymes regulating lipolysis were downregulated in white adipose tissue of these mice. In cultured hepatocytes, selective activation of MC1R reduced ChREBP expression, which is a central transcription factor for lipogenesis. Hepatocyte-specific loss of MC1R disturbs fatty acid metabolism in the liver and leads to an obesity phenotype characterized by enhanced adipocyte hypertrophy and TG accumulation in the liver and circulation.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

International Journal of Obesity
医学-内分泌学与代谢
CiteScore
10.00
自引率
2.00%
发文量
221
审稿时长
3 months
期刊介绍:
The International Journal of Obesity is a multi-disciplinary forum for research describing basic, clinical and applied studies in biochemistry, physiology, genetics and nutrition, molecular, metabolic, psychological and epidemiological aspects of obesity and related disorders.
We publish a range of content types including original research articles, technical reports, reviews, correspondence and brief communications that elaborate on significant advances in the field and cover topical issues.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


