通过将光学纯度极高的 4-苯基-2-恶唑烷酮与 3,3,3-三氟-1-硝基苯进行 Aza-Michael 加成,开发安全实用的 (S)- 和 (R)-N-Boc-3-(Trifluoromethyl)piperazines 对映体纯度合成法
IF 3.1
3区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
摘要
(S)-和(R)-N-叔丁氧羰基-3-(三氟甲基)哌嗪是合理设计药物的诱人构件。迄今为止,它们的手性合成方法尚不为人知。通过手性 3,3,3-三氟丙烷-1,2-二胺的三种合成方法的探索,获得了一条可扩展的路线,并已用于多克合成。由于产量低、纯化繁琐、试剂供应有限以及安全问题,有两条路线无法进行放大。成功且实用的路线依赖于一种改进的工艺,以减轻 (E)-3,3,3- 三氟-1-硝基丙-1-烯合成的安全问题,该工艺被用作高非对映选择性杂-迈克尔加成光学纯 4-苯基-2-噁唑烷酮的储备溶液。通过 Boc 保护氨基,可以在温和的条件下将其转化为手性 N-Boc 保护的 3,3,3-三氟丙烷-1,2-二胺,而无需使用手性色谱法。用 2-氯乙酰氯酰胺化手性 N-叔丁氧羰基保护的 3,3,3-三氟丙烷-1,2-二胺,然后进行分子内环化和还原,可得到对映体纯的 N-叔丁氧羰基-3-(三氟甲基)哌嗪。本文章由计算机程序翻译,如有差异,请以英文原文为准。
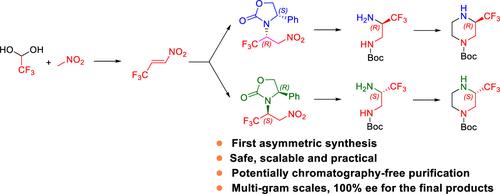
Development of a Safe and Practical Synthesis of Enantiomerically Pure (S)- and (R)-N-Boc-3-(Trifluoromethyl)piperazines Enabled by Aza-Michael Addition of Optically Pure 4-Phenyl-2-Oxazolidinone to 3,3,3-Trifluoro-1-Nitropropene
(S)- and (R)-N-Boc-3-(trifluoromethyl)piperazines are attractive building blocks for the rational design of drugs. Until now, their chiral syntheses were unknown. Exploration of three synthetic approaches via chiral 3,3,3-trifluoropropane-1,2-diamines yielded a scalable route that has been used for multigram synthesis. Two routes were not amendable for scale-up due to low yielding, tedious purification, limited availability of reagents, and safety issues. The successful and practical route relied on a modified process to mitigate the safety issues for the synthesis of (E)-3,3,3-trifluoro-1-nitroprop-1-ene, which was used as a stock solution for the highly diastereoselective aza-Michael addition of optically pure 4-phenyl-2-oxazolidinone. Boc protection of an amino group allowed subsequent transformations to chiral N-Boc-protected 3,3,3-trifluoropropane-1,2-diamine under mild conditions, without the need for chiral chromatography. The amidation of chiral N-Boc-protected 3,3,3-trifluoropropane-1,2-diamine with 2-chloroacetyl chloride, followed by intramolecular cyclization and subsequent reduction afforded enantiomerically pure N-Boc-3-(trifluoromethyl)piperazines.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
6.90
自引率
14.70%
发文量
251
审稿时长
2 months
期刊介绍:
The journal Organic Process Research & Development serves as a communication tool between industrial chemists and chemists working in universities and research institutes. As such, it reports original work from the broad field of industrial process chemistry but also presents academic results that are relevant, or potentially relevant, to industrial applications. Process chemistry is the science that enables the safe, environmentally benign and ultimately economical manufacturing of organic compounds that are required in larger amounts to help address the needs of society. Consequently, the Journal encompasses every aspect of organic chemistry, including all aspects of catalysis, synthetic methodology development and synthetic strategy exploration, but also includes aspects from analytical and solid-state chemistry and chemical engineering, such as work-up tools,process safety, or flow-chemistry. The goal of development and optimization of chemical reactions and processes is their transfer to a larger scale; original work describing such studies and the actual implementation on scale is highly relevant to the journal. However, studies on new developments from either industry, research institutes or academia that have not yet been demonstrated on scale, but where an industrial utility can be expected and where the study has addressed important prerequisites for a scale-up and has given confidence into the reliability and practicality of the chemistry, also serve the mission of OPR&D as a communication tool between the different contributors to the field.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


